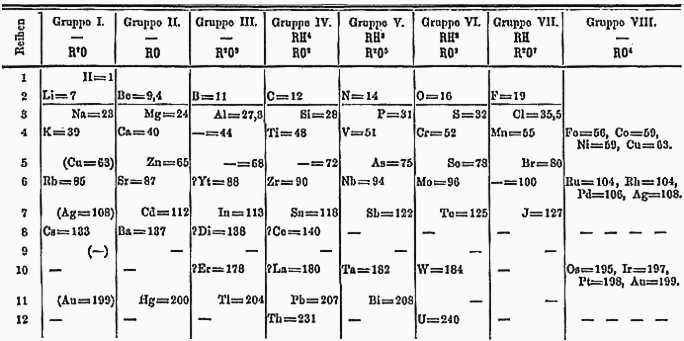
The history of chemistry represents a time span from
ancient history
Ancient history is a time period from the beginning of writing and recorded human history to as far as late antiquity. The span of recorded history is roughly 5,000 years, beginning with the Sumerian cuneiform script. Ancient history cove ...
to the present. By 1000 BC, civilizations used technologies that would eventually form the basis of the various branches of chemistry. Examples include the discovery of fire, extracting
metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
s from
ore
Ore is natural rock or sediment that contains one or more valuable minerals, typically containing metals, that can be mined, treated and sold at a profit.Encyclopædia Britannica. "Ore". Encyclopædia Britannica Online. Retrieved 7 Apr ...
s, making
pottery
Pottery is the process and the products of forming vessels and other objects with clay and other ceramic materials, which are fired at high temperatures to give them a hard and durable form. Major types include earthenware, stoneware and por ...
and glazes, fermenting
beer
Beer is one of the oldest and the most widely consumed type of alcoholic drink in the world, and the third most popular drink overall after water and tea. It is produced by the brewing and fermentation of starches, mainly derived from ce ...
and
wine
Wine is an alcoholic drink typically made from fermented grapes. Yeast consumes the sugar in the grapes and converts it to ethanol and carbon dioxide, releasing heat in the process. Different varieties of grapes and strains of yeasts are m ...
, extracting chemicals from plants for
medicine
Medicine is the science and practice of caring for a patient, managing the diagnosis, prognosis, prevention, treatment, palliation of their injury or disease, and promoting their health. Medicine encompasses a variety of health care pract ...
and
perfume
Perfume (, ; french: parfum) is a mixture of fragrant essential oils or aroma compounds (fragrances), fixatives and solvents, usually in liquid form, used to give the human body, animals, food, objects, and living-spaces an agreeable scent. Th ...
, rendering fat into
soap
Soap is a salt of a fatty acid used in a variety of cleansing and lubricating products. In a domestic setting, soaps are surfactants usually used for washing, bathing, and other types of housekeeping. In industrial settings, soaps are use ...
, making
glass
Glass is a non-crystalline, often transparent, amorphous solid that has widespread practical, technological, and decorative use in, for example, window panes, tableware, and optics. Glass is most often formed by rapid cooling (quenching) of ...
,
and making
alloy
An alloy is a mixture of chemical elements of which at least one is a metal. Unlike chemical compounds with metallic bases, an alloy will retain all the properties of a metal in the resulting material, such as electrical conductivity, ductility, ...
s like
bronze
Bronze is an alloy consisting primarily of copper, commonly with about 12–12.5% tin and often with the addition of other metals (including aluminium, manganese, nickel, or zinc) and sometimes non-metals, such as phosphorus, or metalloids such ...
.
The protoscience of chemistry,
alchemy
Alchemy (from Arabic: ''al-kīmiyā''; from Ancient Greek: χυμεία, ''khumeía'') is an ancient branch of natural philosophy, a philosophical and protoscientific tradition that was historically practiced in China, India, the Muslim world, ...
, was unsuccessful in explaining the nature of matter and its transformations. However, by performing experiments and recording the results, alchemists set the stage for modern chemistry. While both
alchemy
Alchemy (from Arabic: ''al-kīmiyā''; from Ancient Greek: χυμεία, ''khumeía'') is an ancient branch of natural philosophy, a philosophical and protoscientific tradition that was historically practiced in China, India, the Muslim world, ...
and
chemistry
Chemistry is the science, scientific study of the properties and behavior of matter. It is a natural science that covers the Chemical element, elements that make up matter to the chemical compound, compounds made of atoms, molecules and ions ...
are concerned with matter and its transformations,
chemist
A chemist (from Greek ''chēm(ía)'' alchemy; replacing ''chymist'' from Medieval Latin ''alchemist'') is a scientist trained in the study of chemistry. Chemists study the composition of matter and its properties. Chemists carefully describe th ...
s are seen as applying
scientific method
The scientific method is an empirical method for acquiring knowledge that has characterized the development of science since at least the 17th century (with notable practitioners in previous centuries; see the article history of scientific m ...
to their work.
The history of chemistry is intertwined with the
history of thermodynamics
The history of thermodynamics is a fundamental strand in the history of physics, the history of chemistry, and the history of science in general. Owing to the relevance of thermodynamics in much of science and technology, its history is finely wo ...
, especially through the work of
Willard Gibbs
Josiah Willard Gibbs (; February 11, 1839 – April 28, 1903) was an American scientist who made significant theoretical contributions to physics, chemistry, and mathematics. His work on the applications of thermodynamics was instrumental in t ...
.
Ancient history
Early humans
A 100,000-year-old
ochre
Ochre ( ; , ), or ocher in American English, is a natural clay earth pigment, a mixture of ferric oxide and varying amounts of clay and sand. It ranges in colour from yellow to deep orange or brown. It is also the name of the colours produced ...
-processing workshop was found at
Blombos Cave in
South Africa
South Africa, officially the Republic of South Africa (RSA), is the southernmost country in Africa. It is bounded to the south by of coastline that stretch along the South Atlantic and Indian Oceans; to the north by the neighbouring countri ...
. It indicates that early humans had an elementary knowledge of chemistry. Paintings drawn by early humans consisting of early humans mixing animal blood with other liquids found on cave walls also indicate a small knowledge of chemistry.
Early metallurgy
The earliest recorded metal employed by humans seems to be
gold
Gold is a chemical element with the symbol Au (from la, aurum) and atomic number 79. This makes it one of the higher atomic number elements that occur naturally. It is a bright, slightly orange-yellow, dense, soft, malleable, and ductile met ...
, which can be found free or "native". Small amounts of natural gold have been found in Spanish caves used during the late
Paleolithic
The Paleolithic or Palaeolithic (), also called the Old Stone Age (from Greek: παλαιός ''palaios'', "old" and λίθος ''lithos'', "stone"), is a period in human prehistory that is distinguished by the original development of stone too ...
period, around 40,000 BC.
Silver
Silver is a chemical element with the Symbol (chemistry), symbol Ag (from the Latin ', derived from the Proto-Indo-European wikt:Reconstruction:Proto-Indo-European/h₂erǵ-, ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, whi ...
,
copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
,
tin
Tin is a chemical element with the symbol Sn (from la, stannum) and atomic number 50. Tin is a silvery-coloured metal.
Tin is soft enough to be cut with little force and a bar of tin can be bent by hand with little effort. When bent, t ...
and
meteoric iron
Meteoric iron, sometimes meteoritic iron, is a native metal and early-universe protoplanetary-disk remnant found in meteorites and made from the elements iron and nickel, mainly in the form of the mineral phases kamacite and taenite. Meteoric iro ...
can also be found native, allowing a limited amount of
metalworking
Metalworking is the process of shaping and reshaping metals to create useful objects, parts, assemblies, and large scale structures. As a term it covers a wide and diverse range of processes, skills, and tools for producing objects on every scale ...
in ancient cultures.
[Photos, E., 'The Question of Meteorictic versus Smelted Nickel-Rich Iron: Archaeological Evidence and Experimental Results' ''World Archaeology'' Vol. 20, No. 3, Archaeometallurgy (February 1989), pp. 403–421]
Online version
accessed on 2010-02-08. Egyptian weapons made from meteoric iron in about 3000 BC were highly prized as "daggers from Heaven".
[W. Keller (1963) ''The Bible as History'', p. 156 ]
Arguably the first chemical reaction used in a controlled manner was
fire
Fire is the rapid oxidation of a material (the fuel) in the exothermic chemical process of combustion, releasing heat, light, and various reaction Product (chemistry), products.
At a certain point in the combustion reaction, called the ignition ...
. However, for millennia fire was seen simply as a mystical force that could transform one substance into another (burning wood, or boiling water) while producing heat and light. Fire affected many aspects of early societies. These ranged from the simplest facets of everyday life, such as cooking and habitat heating and lighting, to more advanced uses, such as making pottery and bricks and melting of metals to make tools.
It was fire that led to the discovery of
glass
Glass is a non-crystalline, often transparent, amorphous solid that has widespread practical, technological, and decorative use in, for example, window panes, tableware, and optics. Glass is most often formed by rapid cooling (quenching) of ...
and the
purification of metals; this was followed by the rise of
metallurgy
Metallurgy is a domain of materials science and engineering that studies the physical and chemical behavior of metallic elements, their inter-metallic compounds, and their mixtures, which are known as alloys.
Metallurgy encompasses both the sc ...
. During the early stages of metallurgy, methods of purification of metals were sought, and gold, known in
ancient Egypt as early as 2900 BC, became a precious metal.
Bronze Age
Certain metals can be recovered from their ores by simply heating the rocks in a fire: notably
tin
Tin is a chemical element with the symbol Sn (from la, stannum) and atomic number 50. Tin is a silvery-coloured metal.
Tin is soft enough to be cut with little force and a bar of tin can be bent by hand with little effort. When bent, t ...
,
lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cu ...
and (at a higher temperature) copper. This process is known as
smelting
Smelting is a process of applying heat to ore, to extract a base metal. It is a form of extractive metallurgy. It is used to extract many metals from their ores, including silver, iron, copper, and other base metals. Smelting uses heat and a ch ...
. The first evidence of this extractive metallurgy dates from the 6th and 5th millennia BC, and was found in the archaeological sites of the
Vinča culture
The Vinča culture (), also known as Turdaș culture or Turdaș–Vinča culture, is a Neolithic archaeological culture of Southeast Europe, dated to the period 5700–4500 BC or 5300–4700/4500 BC.. Named for its type site, Vinča-Belo Brdo ...
,
Majdanpek
Majdanpek ( sr-cyr, Мајданпек; ro, Maidan) is a town and municipality located in the Bor District of the Southern and Eastern Serbia, eastern Serbia, and is not far from the border of Romania. According to 2011 census, the municipality ...
,
Jarmovac
Priboj ( sr-Cyrl, Прибој, ) is a town and municipality located in the Zlatibor District of southwestern Serbia. The population of the town is 14,920, while the population of the municipality is 27,133.
Geography
The municipality of Priboj i ...
and
Pločnik in
Serbia
Serbia (, ; Serbian language, Serbian: , , ), officially the Republic of Serbia (Serbian language, Serbian: , , ), is a landlocked country in Southeast Europe, Southeastern and Central Europe, situated at the crossroads of the Pannonian Bas ...
. To date, the earliest copper smelting is found at the Belovode site; these examples include a copper axe from 5500 BC. Other signs of early metals are found from the third millennium BC in places like
Palmela
Palmela () is a town and a municipality in Portugal. The population in 2011 was 62,831, in an area of 465.12 km².
The municipality is located in the Lisboa Region and Setúbal District, about south of Lisbon. The municipal holiday is 1 Ju ...
(Portugal),
Los Millares
Los Millares is a Chalcolithic occupation site 17 km north of Almería, in the municipality of Santa Fe de Mondújar, Andalucía, Spain. The complex was in use from the end of the fourth millennium (c. 3000 BC) to the end of the third mi ...
(Spain), and
Stonehenge
Stonehenge is a prehistoric monument on Salisbury Plain in Wiltshire, England, west of Amesbury. It consists of an outer ring of vertical sarsen standing stones, each around high, wide, and weighing around 25 tons, topped by connectin ...
(United Kingdom). However, as often happens in the study of
prehistoric
Prehistory, also known as pre-literary history, is the period of human history between the use of the first stone tools by hominins 3.3 million years ago and the beginning of recorded history with the invention of writing systems. The use of ...
times, the ultimate beginnings cannot be clearly defined and new discoveries are ongoing.

These first metals were single elements, or else combinations as naturally occurred. By combining copper and tin, a superior metal could be made, an
alloy
An alloy is a mixture of chemical elements of which at least one is a metal. Unlike chemical compounds with metallic bases, an alloy will retain all the properties of a metal in the resulting material, such as electrical conductivity, ductility, ...
called
bronze
Bronze is an alloy consisting primarily of copper, commonly with about 12–12.5% tin and often with the addition of other metals (including aluminium, manganese, nickel, or zinc) and sometimes non-metals, such as phosphorus, or metalloids such ...
. This was a major technological shift that began the
Bronze Age
The Bronze Age is a historic period, lasting approximately from 3300 BC to 1200 BC, characterized by the use of bronze, the presence of writing in some areas, and other early features of urban civilization. The Bronze Age is the second pri ...
about 3500 BC. The Bronze Age was a period in human cultural development when the most advanced metalworking (at least in systematic and widespread use) included techniques for smelting
copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
and
tin
Tin is a chemical element with the symbol Sn (from la, stannum) and atomic number 50. Tin is a silvery-coloured metal.
Tin is soft enough to be cut with little force and a bar of tin can be bent by hand with little effort. When bent, t ...
from naturally occurring outcroppings of copper ores, and then smelting those ores to cast bronze. These naturally occurring ores typically included arsenic as a common impurity. Copper/tin ores are rare, as reflected in the absence of tin bronzes in
western Asia
Western Asia, West Asia, or Southwest Asia, is the westernmost subregion of the larger geographical region of Asia, as defined by some academics, UN bodies and other institutions. It is almost entirely a part of the Middle East, and includes Ana ...
before 3000 BC.
After the Bronze Age, the history of metallurgy was marked by armies seeking better weaponry. States in
Eurasia
Eurasia (, ) is the largest continental area on Earth, comprising all of Europe and Asia. Primarily in the Northern and Eastern Hemispheres, it spans from the British Isles and the Iberian Peninsula in the west to the Japanese archipelago a ...
prospered when they made the superior alloys, which, in turn, made better armor and better weapons. Significant progress in metallurgy and alchemy was made in
ancient India
According to consensus in modern genetics, anatomically modern humans first arrived on the Indian subcontinent from Africa between 73,000 and 55,000 years ago. Quote: "Y-Chromosome and Mt-DNA data support the colonization of South Asia by m ...
.
Iron Age
The extraction of
iron
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in f ...
from its ore into a workable metal is much more difficult than copper or tin. While iron is not better suited for tools than bronze (until
steel
Steel is an alloy made up of iron with added carbon to improve its strength and fracture resistance compared to other forms of iron. Many other elements may be present or added. Stainless steels that are corrosion- and oxidation-resistant ty ...
was discovered), iron ore is much more abundant and common than either copper or tin, and therefore more often available locally, with no need to trade for it.
Iron working appears to have been invented by the
Hittites
The Hittites () were an Anatolian people who played an important role in establishing first a kingdom in Kussara (before 1750 BC), then the Kanesh or Nesha kingdom (c. 1750–1650 BC), and next an empire centered on Hattusa in north-centra ...
in about 1200 BC, beginning the
Iron Age
The Iron Age is the final epoch of the three-age division of the prehistory and protohistory of humanity. It was preceded by the Stone Age (Paleolithic, Mesolithic, Neolithic) and the Bronze Age (Chalcolithic). The concept has been mostly appl ...
. The secret of extracting and working iron was a key factor in the success of the
Philistines
The Philistines ( he, פְּלִשְׁתִּים, Pəlīštīm; Koine Greek (LXX): Φυλιστιείμ, romanized: ''Phulistieím'') were an ancient people who lived on the south coast of Canaan from the 12th century BC until 604 BC, when ...
.
[
The Iron Age refers to the advent of iron working (]ferrous metallurgy
Ferrous metallurgy is the metallurgy of iron and its alloys. The earliest surviving prehistoric iron artifacts, from the 4th millennium BC in Egypt, were made from meteoritic iron-nickel. It is not known when or where the smelting of iron from ...
). Historical developments in ferrous metallurgy can be found in a wide variety of past cultures and civilizations. These include the ancient and medieval kingdoms and empires of the Middle East and Near East, ancient Iran
The history of Iran is intertwined with the history of a larger region known as Greater Iran, comprising the area from Anatolia in the west to the borders of Ancient India and the Syr Darya in the east, and from the Caucasus and the Eurasian S ...
, ancient Egypt, ancient Nubia
Nubia () (Nobiin: Nobīn, ) is a region along the Nile river encompassing the area between the first cataract of the Nile (just south of Aswan in southern Egypt) and the confluence of the Blue and White Niles (in Khartoum in central Sudan), or ...
, and Anatolia
Anatolia, tr, Anadolu Yarımadası), and the Anatolian plateau, also known as Asia Minor, is a large peninsula in Western Asia and the westernmost protrusion of the Asian continent. It constitutes the major part of modern-day Turkey. The re ...
(Turkey), Ancient Nok, Carthage
Carthage was the capital city of Ancient Carthage, on the eastern side of the Lake of Tunis in what is now Tunisia. Carthage was one of the most important trading hubs of the Ancient Mediterranean and one of the most affluent cities of the classi ...
, the Greeks
The Greeks or Hellenes (; el, Έλληνες, ''Éllines'' ) are an ethnic group and nation indigenous to the Eastern Mediterranean and the Black Sea regions, namely Greece, Cyprus, Albania, Italy, Turkey, Egypt, and, to a lesser extent, oth ...
and Roman
Roman or Romans most often refers to:
*Rome, the capital city of Italy
*Ancient Rome, Roman civilization from 8th century BC to 5th century AD
*Roman people, the people of ancient Rome
*''Epistle to the Romans'', shortened to ''Romans'', a letter ...
s of ancient Europe, medieval Europe, ancient and medieval China, ancient and medieval India, ancient and medieval Japan, amongst others. Many applications, practices, and devices associated with or involved in metallurgy were established in ancient China, such as the innovation of the blast furnace
A blast furnace is a type of metallurgical furnace used for smelting to produce industrial metals, generally pig iron, but also others such as lead or copper. ''Blast'' refers to the combustion air being "forced" or supplied above atmospheric ...
, cast iron
Cast iron is a class of iron–carbon alloys with a carbon content more than 2%. Its usefulness derives from its relatively low melting temperature. The alloy constituents affect its color when fractured: white cast iron has carbide impuriti ...
, hydraulic
Hydraulics (from Greek: Υδραυλική) is a technology and applied science using engineering, chemistry, and other sciences involving the mechanical properties and use of liquids. At a very basic level, hydraulics is the liquid counter ...
-powered trip hammer
A trip hammer, also known as a tilt hammer or helve hammer, is a massive powered hammer. Traditional uses of trip hammers include pounding, decorticating and polishing of grain in agriculture. In mining, trip hammers were used for crushing meta ...
s, and double-acting piston bellows
A bellows or pair of bellows is a device constructed to furnish a strong blast of air. The simplest type consists of a flexible bag comprising a pair of rigid boards with handles joined by flexible leather sides enclosing an approximately airtigh ...
.[Temple, Robert K.G. (2007). ''The Genius of China: 3,000 Years of Science, Discovery, and Invention'' (3rd edition). London: ]André Deutsch
André Deutsch (15 November 1917 – 11 April 2000) was a Hungarian-born British publisher who founded an eponymous publishing company in 1951.
Biography
Deutsch was born on 15 November 1917 in Budapest, Hungary, the son of a Jewish dentis ...
. pp. 44–56. .
Classical antiquity and atomism
 Philosophical attempts to rationalize why different substances have different properties (color, density, smell), exist in different states (gaseous, liquid, and solid), and react in a different manner when exposed to environments, for example to water or fire or temperature changes, led ancient philosophers to postulate the first theories on nature and chemistry. The history of such philosophical theories that relate to chemistry can probably be traced back to every single ancient civilization. The common aspect in all these theories was the attempt to identify a small number of primary
Philosophical attempts to rationalize why different substances have different properties (color, density, smell), exist in different states (gaseous, liquid, and solid), and react in a different manner when exposed to environments, for example to water or fire or temperature changes, led ancient philosophers to postulate the first theories on nature and chemistry. The history of such philosophical theories that relate to chemistry can probably be traced back to every single ancient civilization. The common aspect in all these theories was the attempt to identify a small number of primary classical elements
Classical elements typically refer to earth, water, air, fire, and (later) aether which were proposed to explain the nature and complexity of all matter in terms of simpler substances. Ancient cultures in Greece, Tibet, and India had simil ...
that make up all the various substances in nature. Substances like air, water, and soil/earth, energy forms, such as fire and light, and more abstract concepts such as thoughts, aether, and heaven, were common in ancient civilizations even in the absence of any cross-fertilization: for example ancient Greek, Indian, Mayan, and Chinese philosophies all considered air
The atmosphere of Earth is the layer of gases, known collectively as air, retained by Earth's gravity that surrounds the planet and forms its planetary atmosphere. The atmosphere of Earth protects life on Earth by creating pressure allowing f ...
, water
Water (chemical formula ) is an inorganic, transparent, tasteless, odorless, and nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a ...
, earth
Earth is the third planet from the Sun and the only astronomical object known to harbor life. While large volumes of water can be found throughout the Solar System, only Earth sustains liquid surface water. About 71% of Earth's surfa ...
and fire
Fire is the rapid oxidation of a material (the fuel) in the exothermic chemical process of combustion, releasing heat, light, and various reaction Product (chemistry), products.
At a certain point in the combustion reaction, called the ignition ...
as primary elements.
Ancient world
Around 420 BC, Empedocles
Empedocles (; grc-gre, Ἐμπεδοκλῆς; , 444–443 BC) was a Greek pre-Socratic philosopher and a native citizen of Akragas, a Greek city in Sicily. Empedocles' philosophy is best known for originating the cosmogonic theory of the fo ...
stated that all matter is made up of four elemental substances: earth, fire, air and water. The early theory of atomism
Atomism (from Greek , ''atomon'', i.e. "uncuttable, indivisible") is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms.
References to the concept of atomism and its atoms ...
can be traced back to ancient Greece
Ancient Greece ( el, Ἑλλάς, Hellás) was a northeastern Mediterranean civilization, existing from the Greek Dark Ages of the 12th–9th centuries BC to the end of classical antiquity ( AD 600), that comprised a loose collection of cult ...
and ancient India
According to consensus in modern genetics, anatomically modern humans first arrived on the Indian subcontinent from Africa between 73,000 and 55,000 years ago. Quote: "Y-Chromosome and Mt-DNA data support the colonization of South Asia by m ...
.Will Durant
William James Durant (; November 5, 1885 – November 7, 1981) was an American writer, historian, and philosopher. He became best known for his work '' The Story of Civilization'', which contains 11 volumes and details the history of eastern a ...
(1935), ''Our Oriental Heritage'':
Democritus
Democritus (; el, Δημόκριτος, ''Dēmókritos'', meaning "chosen of the people"; – ) was an Ancient Greek pre-Socratic philosopher from Abdera, primarily remembered today for his formulation of an atomic theory of the universe. No ...
, who declared that matter is composed of indivisible and indestructible particles called "atomos" around 380 BC. Earlier, Leucippus
Leucippus (; el, Λεύκιππος, ''Leúkippos''; fl. 5th century BCE) is a pre-Socratic Greek philosopher who has been credited as the first philosopher to develop a theory of atomism.
Leucippus' reputation, even in antiquity, was obscured ...
also declared that atoms were the most indivisible part of matter (this coincided with a similar declaration by Indian
Indian or Indians may refer to:
Peoples South Asia
* Indian people, people of Indian nationality, or people who have an Indian ancestor
** Non-resident Indian, a citizen of India who has temporarily emigrated to another country
* South Asia ...
philosopher Kanada Kanada may refer to:
*Kanada (philosopher), the Hindu sage who founded the philosophy of Vaisheshika
*Kanada (family of ragas), a group of ragas in Hindustani music
*Kanada (surname)
*Kanada Station, train station in Fukuoka, Japan
*Kannada, one of ...
in his Vaisheshika
Vaisheshika or Vaiśeṣika ( sa, वैशेषिक) is one of the six schools of Indian philosophy (Vedic systems) from ancient India. In its early stages, the Vaiśeṣika was an independent philosophy with its own metaphysics, epistemolog ...
sutra
''Sutra'' ( sa, सूत्र, translit=sūtra, translit-std=IAST, translation=string, thread)Monier Williams, ''Sanskrit English Dictionary'', Oxford University Press, Entry fo''sutra'' page 1241 in Indian literary traditions refers to an aph ...
s around the same time period).[ ]Aristotle
Aristotle (; grc-gre, Ἀριστοτέλης ''Aristotélēs'', ; 384–322 BC) was a Greek philosopher and polymath during the Classical period in Ancient Greece. Taught by Plato, he was the founder of the Peripatetic school of phil ...
opposed the existence of atoms in 330 BC. A Greek text attributed to Polybus the physician (ca. 380 BC) argued that the human body is composed of four humours
Humorism, the humoral theory, or humoralism, was a system of medicine detailing a supposed makeup and workings of the human body, adopted by Ancient Greek and Roman physicians and philosophers.
Humorism began to fall out of favor in the 1850s ...
instead. Epicurus
Epicurus (; grc-gre, Ἐπίκουρος ; 341–270 BC) was an ancient Greek philosopher and sage who founded Epicureanism, a highly influential school of philosophy. He was born on the Greek island of Samos to Athenian parents. Influenced ...
(fl. 300 BC) postulated a universe of indestructible atoms in which man himself is responsible for achieving a balanced life.
With the goal of explaining Epicurean philosophy
Epicureanism is a system of philosophy founded around 307 BC based upon the teachings of the ancient Greek philosopher Epicurus. Epicureanism was originally a challenge to Platonism. Later its main opponent became Stoicism.
Few writings by Epi ...
to a Roman audience, the Roman
Roman or Romans most often refers to:
*Rome, the capital city of Italy
*Ancient Rome, Roman civilization from 8th century BC to 5th century AD
*Roman people, the people of ancient Rome
*''Epistle to the Romans'', shortened to ''Romans'', a letter ...
poet and philosopher Lucretius
Titus Lucretius Carus ( , ; – ) was a Roman poet and philosopher. His only known work is the philosophical poem ''De rerum natura'', a didactic work about the tenets and philosophy of Epicureanism, and which usually is translated into E ...
wrote ''De rerum natura
''De rerum natura'' (; ''On the Nature of Things'') is a first-century BC didactic poem by the Roman poet and philosopher Lucretius ( – c. 55 BC) with the goal of explaining Epicurean philosophy to a Roman audience. The poem, written in some 7 ...
'' (The Nature of Things) in 50 BC. In the work, Lucretius presents the principles of atomism
Atomism (from Greek , ''atomon'', i.e. "uncuttable, indivisible") is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms.
References to the concept of atomism and its atoms ...
; the nature of the mind
The mind is the set of faculties responsible for all mental phenomena. Often the term is also identified with the phenomena themselves. These faculties include thought, imagination, memory, will, and sensation. They are responsible for various m ...
and soul
In many religious and philosophical traditions, there is a belief that a soul is "the immaterial aspect or essence of a human being".
Etymology
The Modern English noun ''soul'' is derived from Old English ''sāwol, sāwel''. The earliest attes ...
; explanations of sensation
Sensation (psychology) refers to the processing of the senses by the sensory system.
Sensation or sensations may also refer to:
In arts and entertainment In literature
*Sensation (fiction), a fiction writing mode
*Sensation novel, a British ...
and thought; the development of the world and its phenomena; and explains a variety of celestial
Celestial may refer to:
Science
* Objects or events seen in the sky and the following astronomical terms:
** Astronomical object, a naturally occurring physical entity, association, or structure that exists in the observable universe
** Celes ...
and terrestrial
Terrestrial refers to things related to land or the planet Earth.
Terrestrial may also refer to:
* Terrestrial animal, an animal that lives on land opposed to living in water, or sometimes an animal that lives on or near the ground, as opposed to ...
phenomena.
The earliest alchemists in the Western tradition seemed to have come from Greco-Roman Egypt
The history of Egypt has been long and wealthy, due to the flow of the Nile River with its fertile banks and delta, as well as the accomplishments of Egypt's native inhabitants and outside influence. Much of Egypt's ancient history was a myste ...
in the first centuries AD. In addition to technical work, many of them invented chemical apparatuses. The ''bain-marie'', or water bath, is named for Mary the Jewess
Mary or Maria the Jewess ( la, Maria Hebraea), also known as Mary the Prophetess ( la, Maria Prophetissa) or Maria the Copt ( ar, مارية القبطية, Māriyya al-Qibṭiyya), was an early alchemist known from the works of Zosimos of Panopo ...
. Her work also gives the first descriptions of the ''tribikos'' and ''kerotakis''. Cleopatra the Alchemist
Cleopatra the Alchemist (Greek: Κλεοπάτρα; fl. c. 3rd century AD) was a Greek alchemist, author, and philosopher. She experimented with practical alchemy but is also credited as one of the four female alchemists who could produce the P ...
described furnaces and has been credited with the invention of the alembic
An alembic (from ar, الإنبيق, al-inbīq, originating from grc, ἄμβιξ, ambix, 'cup, beaker') is an alchemical still consisting of two vessels connected by a tube, used for distillation of liquids.
Description
The complete disti ...
. Later, Zosimos of Panopolis
Zosimos of Panopolis ( el, Ζώσιμος ὁ Πανοπολίτης; also known by the Latin name Zosimus Alchemista, i.e. "Zosimus the Alchemist") was a Greco-Egyptian alchemist and Gnostic mystic who lived at the end of the 3rd and beginning ...
wrote books on alchemy, which he called ''cheirokmeta'', the Greek word for "things made by hand." These works include many references to recipes and procedures, as well as descriptions of instruments. Much of the early development of purification methods were described earlier by Pliny the Elder
Gaius Plinius Secundus (AD 23/2479), called Pliny the Elder (), was a Roman author, naturalist and natural philosopher, and naval and army commander of the early Roman Empire, and a friend of the emperor Vespasian. He wrote the encyclopedic '' ...
in his Naturalis Historia
The ''Natural History'' ( la, Naturalis historia) is a work by Pliny the Elder. The largest single work to have survived from the Roman Empire to the modern day, the ''Natural History'' compiles information gleaned from other ancient authors. ...
. He tried to explain those methods, as well as making acute observations of the state of many minerals.
Medieval alchemy
 The elemental system used in medieval
The elemental system used in medieval alchemy
Alchemy (from Arabic: ''al-kīmiyā''; from Ancient Greek: χυμεία, ''khumeía'') is an ancient branch of natural philosophy, a philosophical and protoscientific tradition that was historically practiced in China, India, the Muslim world, ...
was developed primarily by the Persian
Persian may refer to:
* People and things from Iran, historically called ''Persia'' in the English language
** Persians, the majority ethnic group in Iran, not to be conflated with the Iranic peoples
** Persian language, an Iranian language of the ...
-Arab
The Arabs (singular: Arab; singular ar, عَرَبِيٌّ, DIN 31635: , , plural ar, عَرَب, DIN 31635: , Arabic pronunciation: ), also known as the Arab people, are an ethnic group mainly inhabiting the Arab world in Western Asia, ...
alchemist Jābir ibn Hayyān
Abū Mūsā Jābir ibn Ḥayyān (Arabic: , variously called al-Ṣūfī, al-Azdī, al-Kūfī, or al-Ṭūsī), died 806−816, is the purported author of an enormous number and variety of works in Arabic, often called the Jabirian corpus. The ...
and was rooted in the classical elements of Greek tradition. His system consisted of the four Aristotelian elements of air, earth, fire, and water in addition to two philosophical elements: sulphur
Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula ...
, characterizing the principle of combustibility, "the stone which burns"; and mercury
Mercury commonly refers to:
* Mercury (planet), the nearest planet to the Sun
* Mercury (element), a metallic chemical element with the symbol Hg
* Mercury (mythology), a Roman god
Mercury or The Mercury may also refer to:
Companies
* Merc ...
, characterizing the principle of metallic properties. They were seen by early alchemists as idealized expressions of irreducible components of the universe
The universe is all of space and time and their contents, including planets, stars, galaxies, and all other forms of matter and energy. The Big Bang theory is the prevailing cosmological description of the development of the universe. Acc ...
and are of larger consideration within philosophical alchemy.
The three metallic principles (sulphur to flammability or combustion, mercury to volatility and stability, and salt
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quantitie ...
to solidity) became the ''tria prima'' of the Swiss alchemist Paracelsus
Paracelsus (; ; 1493 – 24 September 1541), born Theophrastus von Hohenheim (full name Philippus Aureolus Theophrastus Bombastus von Hohenheim), was a Swiss physician, alchemist, lay theologian, and philosopher of the German Renaissance.
He w ...
. He reasoned that Aristotle's four-element theory appeared in bodies as three principles. Paracelsus saw these principles as fundamental and justified them by recourse to the description of how wood burns in fire. Mercury included the cohesive principle, so that when it left the wood (in smoke) the wood fell apart. Smoke described the volatility (the mercurial principle), the heat-giving flames described flammability (sulphur), and the remnant ash described solidity (salt).
The philosopher's stone
 Alchemy is defined by the
Alchemy is defined by the Hermetic
Hermetic or related forms may refer to:
* of or related to the ancient Greek Olympian god Hermes
* of or related to Hermes Trismegistus, a legendary Hellenistic figure based on the Greek god Hermes and the Egyptian god Thoth
** , the ancient and m ...
quest for the philosopher's stone
The philosopher's stone or more properly philosophers' stone (Arabic: حجر الفلاسفة, , la, lapis philosophorum), is a mythic alchemical substance capable of turning base metals such as mercury into gold (, from the Greek , "gold", a ...
, the study of which is steeped in symbolic mysticism, and differs greatly from modern science. Alchemists toiled to make transformations on an esoteric
Western esotericism, also known as esotericism, esoterism, and sometimes the Western mystery tradition, is a term scholars use to categorise a wide range of loosely related ideas and movements that developed within Western society. These ideas a ...
(spiritual) and/or exoteric
Exoteric refers to knowledge that is outside and independent from a person's experience and can be ascertained by anyone (related to common sense).
The word is derived from the comparative form of Greek ἔξω ''eksô'', "from, out of, outside". ...
(practical) level. It was the protoscientific
__NOTOC__
In the philosophy of science, there are several definitions of protoscience. Its simplest meaning (most closely reflecting its roots of ''wikt:proto-#Prefix, proto-'' + ''science'') involves the earliest eras of the history of science, wh ...
, exoteric aspects of alchemy that contributed heavily to the evolution of chemistry in Greco-Roman Egypt
The history of Egypt has been long and wealthy, due to the flow of the Nile River with its fertile banks and delta, as well as the accomplishments of Egypt's native inhabitants and outside influence. Much of Egypt's ancient history was a myste ...
, in the Islamic Golden Age
The Islamic Golden Age was a period of cultural, economic, and scientific flourishing in the history of Islam, traditionally dated from the 8th century to the 14th century. This period is traditionally understood to have begun during the reign ...
, and then in Europe. Alchemy and chemistry share an interest in the composition and properties of matter, and until the 18th century they were not separate disciplines. The term ''chymistry'' has been used to describe the blend of alchemy and chemistry that existed before that time.
During the Renaissance, exoteric alchemy remained popular in the form of Paracelsian
Paracelsianism (also Paracelsism; German: ') was an early modern History of medicine, medical movement based on the theories and therapies of Paracelsus.
It developed in the second half of the 16th century, during the decades following Paracelsu ...
iatrochemistry
Iatrochemistry (; also known as chemiatria or chemical medicine) is a branch of both chemistry and medicine. Having its roots in alchemy, iatrochemistry seeks to provide chemical solutions to diseases and medical ailments.
This area of science h ...
, while spiritual alchemy flourished, realigned to its Platonic
Plato's influence on Western culture was so profound that several different concepts are linked by being called Platonic or Platonist, for accepting some assumptions of Platonism, but which do not imply acceptance of that philosophy as a whole. It ...
, Hermetic, and Gnostic
Gnosticism (from grc, γνωστικός, gnōstikós, , 'having knowledge') is a collection of religious ideas and systems which coalesced in the late 1st century AD among Jewish and early Christian sects. These various groups emphasized pe ...
roots. Consequently, the symbolic quest for the philosopher's stone was not superseded by scientific advances, and was still the domain of respected scientists and doctors until the early 18th century. Early modern alchemists who are renowned for their scientific contributions include Jan Baptist van Helmont
Jan Baptist van Helmont (; ; 12 January 1580 – 30 December 1644) was a chemist, physiologist, and physician from Brussels. He worked during the years just after Paracelsus and the rise of iatrochemistry, and is sometimes considered to ...
, Robert Boyle
Robert Boyle (; 25 January 1627 – 31 December 1691) was an Anglo-Irish natural philosopher, chemist, physicist, alchemist and inventor. Boyle is largely regarded today as the first modern chemist, and therefore one of the founders of ...
, and Isaac Newton
Sir Isaac Newton (25 December 1642 – 20 March 1726/27) was an English mathematician, physicist, astronomer, alchemist, theologian, and author (described in his time as a "natural philosopher"), widely recognised as one of the grea ...
.
Alchemy in the Islamic world
In the Islamic World
The terms Muslim world and Islamic world commonly refer to the Islamic community, which is also known as the Ummah. This consists of all those who adhere to the religious beliefs and laws of Islam or to societies in which Islam is practiced. In ...
, the Muslim
Muslims ( ar, المسلمون, , ) are people who adhere to Islam, a monotheistic religion belonging to the Abrahamic tradition. They consider the Quran, the foundational religious text of Islam, to be the verbatim word of the God of Abrah ...
s were translating the works of ancient Greek
Greek may refer to:
Greece
Anything of, from, or related to Greece, a country in Southern Europe:
*Greeks, an ethnic group.
*Greek language, a branch of the Indo-European language family.
**Proto-Greek language, the assumed last common ancestor ...
and Hellenistic
In Classical antiquity, the Hellenistic period covers the time in Mediterranean history after Classical Greece, between the death of Alexander the Great in 323 BC and the emergence of the Roman Empire, as signified by the Battle of Actium in ...
philosophers into Arabic and were experimenting with scientific ideas. The Arabic works attributed to the 8th-century alchemist Jābir ibn Hayyān
Abū Mūsā Jābir ibn Ḥayyān (Arabic: , variously called al-Ṣūfī, al-Azdī, al-Kūfī, or al-Ṭūsī), died 806−816, is the purported author of an enormous number and variety of works in Arabic, often called the Jabirian corpus. The ...
introduced a systematic classification of chemical substances, and provided instructions for deriving an inorganic compound (sal ammoniac
Salammoniac, also sal ammoniac or salmiac, is a rare naturally occurring mineral composed of ammonium chloride, NH4Cl. It forms colorless, white, or yellow-brown crystals in the isometric-hexoctahedral class. It has very poor cleavage and is ...
or ammonium chloride) from organic substances (such as plants, blood, and hair) by chemical means. Some Arabic Jabirian works (e.g., the "Book of Mercy", and the "Book of Seventy") were later translated into Latin under the Latinized name "Geber", and in 13th-century Europe an anonymous writer, usually referred to as pseudo-Geber
Pseudo-Geber (or "Latin pseudo-Geber") is the presumed author or group of authors responsible for a corpus of pseudepigraphic alchemical writings dating to the late 13th and early 14th centuries. These writings were falsely attributed to Jabir ...
, started to produce alchemical and metallurgical writings under this name. Later influential Muslim philosophers, such as Abū al-Rayhān al-Bīrūnī
Abu Rayhan Muhammad ibn Ahmad al-Biruni (973 – after 1050) commonly known as al-Biruni, was a Khwarazmian Iranian in scholar and polymath during the Islamic Golden Age. He has been called variously the "founder of Indology", "Father of Co ...
and Avicenna
Ibn Sina ( fa, ابن سینا; 980 – June 1037 CE), commonly known in the West as Avicenna (), was a Persian polymath who is regarded as one of the most significant physicians, astronomers, philosophers, and writers of the Islamic G ...
disputed the theories of alchemy, particularly the theory of the transmutation of metals.
Problems encountered with alchemy
There were several problems with alchemy, as seen from today's standpoint. There was no systematic naming scheme for new compounds, and the language was esoteric and vague to the point that the terminologies meant different things to different people. In fact, according to ''The Fontana History of Chemistry'' (Brock, 1992):
The language of alchemy soon developed an arcane and secretive technical vocabulary designed to conceal information from the uninitiated. To a large degree, this language is incomprehensible to us today, though it is apparent that readers of Geoffery Chaucer's Canon's Yeoman's Tale "The Canon's Yeoman's Tale" is one of '' The Canterbury Tales'' by Geoffrey Chaucer.
The Canon and his Yeoman are not mentioned in the General Prologue of The Canterbury Tales, where most of the other pilgrims are described, but they arrive later ...
or audiences of Ben Jonson
Benjamin "Ben" Jonson (c. 11 June 1572 – c. 16 August 1637) was an English playwright and poet. Jonson's artistry exerted a lasting influence upon English poetry and stage comedy. He popularised the comedy of humours; he is best known for t ...
's The Alchemist
An alchemist is a person who practices alchemy.
Alchemist or Alchemyst may also refer to:
Books and stories
* ''The Alchemist'' (novel), the translated title of a 1988 allegorical novel by Paulo Coelho
* ''The Alchemist'' (play), a play by Be ...
were able to construe it sufficiently to laugh at it.
Chaucer's tale exposed the more fraudulent side of alchemy, especially the manufacture of counterfeit gold from cheap substances. Less than a century earlier, Dante Alighieri
Dante Alighieri (; – 14 September 1321), probably baptized Durante di Alighiero degli Alighieri and often referred to as Dante (, ), was an Italian poet, writer and philosopher. His ''Divine Comedy'', originally called (modern Italian: '' ...
also demonstrated an awareness of this fraudulence, causing him to consign all alchemists to the Inferno
Inferno may refer to:
* Hell, an afterlife place of suffering
* Conflagration, a large uncontrolled fire
Film
* ''L'Inferno'', a 1911 Italian film
* Inferno (1953 film), ''Inferno'' (1953 film), a film noir by Roy Ward Baker
* Inferno (1973 fi ...
in his writings. Soon afterwards, in 1317, the Avignon
Avignon (, ; ; oc, Avinhon, label=Provençal dialect, Provençal or , ; la, Avenio) is the Prefectures in France, prefecture of the Vaucluse Departments of France, department in the Provence-Alpes-Côte d'Azur Regions of France, region of So ...
Pope John XXII
Pope John XXII ( la, Ioannes PP. XXII; 1244 – 4 December 1334), born Jacques Duèze (or d'Euse), was head of the Catholic Church from 7 August 1316 to his death in December 1334.
He was the second and longest-reigning Avignon Pope, elected by ...
ordered all alchemists to leave France for making counterfeit money. A law was passed in England in 1403 which made the "multiplication of metals" punishable by death. Despite these and other apparently extreme measures, alchemy did not die. Royalty and privileged classes still sought to discover the philosopher's stone and the elixir of life for themselves.
There was also no agreed-upon scientific method for making experiments reproducible. Indeed, many alchemists included in their methods irrelevant information such as the timing of the tides or the phases of the moon. The esoteric nature and codified vocabulary of alchemy appeared to be more useful in concealing the fact that they could not be sure of very much at all. As early as the 14th century, cracks seemed to grow in the facade of alchemy; and people became sceptical. Clearly, there needed to be a scientific method in which experiments could be repeated by other people, and results needed to be reported in a clear language that laid out both what was known and what was unknown.
17th and 18th centuries: Early chemistry
 Practical attempts to improve the refining of ores and their extraction to smelt metals was an important source of information for early chemists in the 16th century, among them
Practical attempts to improve the refining of ores and their extraction to smelt metals was an important source of information for early chemists in the 16th century, among them Georg Agricola
Georgius Agricola (; born Georg Pawer or Georg Bauer; 24 March 1494 – 21 November 1555) was a German Humanist scholar, mineralogist and metallurgist. Born in the small town of Glauchau, in the Electorate of Saxony of the Holy Roman Emp ...
(1494–1555), who published his great work '' De re metallica'' in 1556. His work describes the highly developed and complex processes of mining metal ores, metal extraction and metallurgy of the time. His approach removed the mysticism associated with the subject, creating the practical base upon which others could build. The work describes the many kinds of furnace used to smelt ore, and stimulated interest in minerals and their composition. It is no coincidence that he gives numerous references to the earlier author, Pliny the Elder and his ''Naturalis Historia''. Agricola has been described as the "father of metallurgy".
In 1605, Sir Francis Bacon
Francis Bacon, 1st Viscount St Alban (; 22 January 1561 – 9 April 1626), also known as Lord Verulam, was an English philosopher and statesman who served as Attorney General and Lord Chancellor of England. Bacon led the advancement of both n ...
published ''The Proficience and Advancement of Learning'', which contains a description of what would later be known as the scientific method
The scientific method is an empirical method for acquiring knowledge that has characterized the development of science since at least the 17th century (with notable practitioners in previous centuries; see the article history of scientific m ...
. In 1605, Michal Sedziwój
Michael Sendivogius (; pl, Michał Sędziwój; 2 February 1566 – 1636) was a Polish alchemist, philosopher, and medical doctor. A pioneer of chemistry, he developed ways of purification and creation of various acids, metals and other ch ...
publishes the alchemical treatise ''A New Light of Alchemy'' which proposed the existence of the "food of life" within air, much later recognized as oxygen. In 1615 Jean Beguin Jean Beguin (1550–1620) was an iatrochemist noted for his 1610 ''Tyrocinium Chymicum'' (Begin Chemistry)Digital edition, which many consider to be one of the first chemistry textbooks. In the 1615 edition of his textbook, Beguin made the first-e ...
published the '' Tyrocinium Chymicum'', an early chemistry textbook, and in it draws the first-ever chemical equation
A chemical equation is the symbolic representation of a chemical reaction in the form of symbols and chemical formulas. The reactant entities are given on the left-hand side and the product entities on the right-hand side with a plus sign between ...
. In 1637 René Descartes
René Descartes ( or ; ; Latinized: Renatus Cartesius; 31 March 1596 – 11 February 1650) was a French philosopher, scientist, and mathematician, widely considered a seminal figure in the emergence of modern philosophy and science. Mathem ...
publishes ''Discours de la méthode
''Discourse on the Method of Rightly Conducting One's Reason and of Seeking Truth in the Sciences'' (french: Discours de la Méthode Pour bien conduire sa raison, et chercher la vérité dans les sciences) is a philosophical and autobiographical ...
'', which contains an outline of the scientific method.
The Dutch chemist Jan Baptist van Helmont
Jan Baptist van Helmont (; ; 12 January 1580 – 30 December 1644) was a chemist, physiologist, and physician from Brussels. He worked during the years just after Paracelsus and the rise of iatrochemistry, and is sometimes considered to ...
's work ''Ortus medicinae'' was published posthumously in 1648; the book is cited by some as a major transitional work between alchemy and chemistry, and as an important influence on Robert Boyle
Robert Boyle (; 25 January 1627 – 31 December 1691) was an Anglo-Irish natural philosopher, chemist, physicist, alchemist and inventor. Boyle is largely regarded today as the first modern chemist, and therefore one of the founders of ...
. The book contains the results of numerous experiments and establishes an early version of the law of conservation of mass. Working during the time just after Paracelsus
Paracelsus (; ; 1493 – 24 September 1541), born Theophrastus von Hohenheim (full name Philippus Aureolus Theophrastus Bombastus von Hohenheim), was a Swiss physician, alchemist, lay theologian, and philosopher of the German Renaissance.
He w ...
and iatrochemistry
Iatrochemistry (; also known as chemiatria or chemical medicine) is a branch of both chemistry and medicine. Having its roots in alchemy, iatrochemistry seeks to provide chemical solutions to diseases and medical ailments.
This area of science h ...
, Jan Baptist van Helmont suggested that there are insubstantial substances other than air and coined a name for them – "gas
Gas is one of the four fundamental states of matter (the others being solid, liquid, and plasma).
A pure gas may be made up of individual atoms (e.g. a noble gas like neon), elemental molecules made from one type of atom (e.g. oxygen), or ...
", from the Greek word ''chaos''. In addition to introducing the word "gas" into the vocabulary of scientists, van Helmont conducted several experiments involving gases. Jan Baptist van Helmont is also remembered today largely for his ideas on spontaneous generation
Spontaneous generation is a superseded scientific theory that held that living creatures could arise from nonliving matter and that such processes were commonplace and regular. It was hypothesized that certain forms, such as fleas, could arise fr ...
and his 5-year tree experiment, as well as being considered the founder of pneumatic chemistry
In the history of science, pneumatic chemistry is an area of scientific research of the seventeenth, eighteenth, and early nineteenth centuries. Important goals of this work were the understanding of the physical properties of gases and how the ...
.
Robert Boyle

 Anglo-Irish chemist
Anglo-Irish chemist Robert Boyle
Robert Boyle (; 25 January 1627 – 31 December 1691) was an Anglo-Irish natural philosopher, chemist, physicist, alchemist and inventor. Boyle is largely regarded today as the first modern chemist, and therefore one of the founders of ...
(1627–1691) is considered to have initiated the gradual separation of chemistry from alchemy. Although skeptical of elements and convinced of alchemy, Boyle played a key part in elevating the "sacred art" as an independent, fundamental and philosophical discipline. He is best known for Boyle's law
Boyle's law, also referred to as the Boyle–Mariotte law, or Mariotte's law (especially in France), is an experimental gas law that describes the relationship between pressure and volume of a confined gas. Boyle's law has been stated as:
The ...
, which he presented in 1662, though he was not the first to discover it.pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and e ...
and volume
Volume is a measure of occupied three-dimensional space. It is often quantified numerically using SI derived units (such as the cubic metre and litre) or by various imperial or US customary units (such as the gallon, quart, cubic inch). The de ...
of a gas, if the temperature is kept constant within a closed system
A closed system is a natural physical system that does not allow transfer of matter in or out of the system, although — in contexts such as physics, chemistry or engineering — the transfer of energy (''e.g.'' as work or heat) is allowed.
In ...
.[Levine, Ira. N. (1978), p12 gives the original definition.]
Boyle is also credited for his landmark publication ''The Sceptical Chymist
''The Sceptical Chymist: or Chymico-Physical Doubts & Paradoxes'' is the title of a book by Robert Boyle, published in London in 1661. In the form of a dialogue, the ''Sceptical Chymist'' presented Boyle's hypothesis that matter consisted of corp ...
'' (1661), which advocated for a rigorous approach to experimentation among chemists. In the work, Boyle questioned some commonly held alchemical theories and argued for practitioners to be more “philosophical” and less commercially focused. He rejected the classical four elements of earth, fire, air, and water, and proposed a mechanistic alternative of atoms and chemical reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the pos ...
s that could be subject to rigorous experiment.
Boyle also tried to purify chemicals to obtain reproducible reactions. He was a vocal proponent of the mechanical philosophy proposed by René Descartes
René Descartes ( or ; ; Latinized: Renatus Cartesius; 31 March 1596 – 11 February 1650) was a French philosopher, scientist, and mathematician, widely considered a seminal figure in the emergence of modern philosophy and science. Mathem ...
to explain and quantify the physical properties and interactions of material substances. Boyle was an atomist, but favoured the word ''corpuscle'' over ''atoms''. He commented that the finest division of matter where the properties are retained is at the level of corpuscles.
Boyle repeated the tree experiment of van Helmont, and was the first to use indicators
Indicator may refer to:
Biology
* Environmental indicator of environmental health (pressures, conditions and responses)
* Ecological indicator of ecosystem health (ecological processes)
* Health indicator, which is used to describe the health o ...
which changed colors with acidity. He also performed numerous investigations with an air pump
An air pump is a pump for pushing air. Examples include a bicycle pump, pumps that are used to aerate an aquarium or a pond via an airstone; a gas compressor used to power a pneumatic tool, air horn or pipe organ; a bellows used to encourage ...
, and noted that the mercury
Mercury commonly refers to:
* Mercury (planet), the nearest planet to the Sun
* Mercury (element), a metallic chemical element with the symbol Hg
* Mercury (mythology), a Roman god
Mercury or The Mercury may also refer to:
Companies
* Merc ...
fell as air was pumped out. He also observed that pumping the air out of a container would extinguish a flame and kill small animals placed inside. Through his works, Boyle helped to lay the foundations for the chemical revolution
The chemical revolution, also called the ''first chemical revolution'', was the early modern reformulation of chemistry that culminated in the law of conservation of mass and the oxygen theory of combustion.
During the 19th and 20th century, thi ...
two centuries later.
Development and dismantling of phlogiston
 In 1702, German chemist
In 1702, German chemist Georg Stahl
Georg Ernst Stahl (22 October 1659 – 24 May 1734) was a German chemist, physician and philosopher. He was a supporter of vitalism, and until the late 18th century his works on phlogiston were accepted as an explanation for chemical processes.K ...
coined the name "phlogiston
The phlogiston theory is a superseded scientific theory that postulated the existence of a fire-like element called phlogiston () contained within combustible bodies and released during combustion. The name comes from the Ancient Greek (''burni ...
" for the substance believed to be released in the process of burning. Around 1735, Swedish chemist Georg Brandt
Georg Brandt (26 June 1694 – 29 April 1768) was a Swedish chemist and mineralogist who discovered cobalt (c. 1735). He was the first person to discover a metal unknown in ancient times. He is also known for exposing fraudulent alchemists operatin ...
analyzed a dark blue pigment found in copper ore. Brandt demonstrated that the pigment contained a new element, later named cobalt
Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, pr ...
. In 1751, a Swedish chemist and pupil of Stahl's named Axel Fredrik Cronstedt
Baron Axel Fredrik Cronstedt (''/kroonstet/'' 23 December 1722 – 19 August 1765) was a Swedish mineralogist and chemist who discovered the element nickel in 1751 as a mining expert with the Bureau of Mines.
Cronstedt is considered a founder of ...
, identified an impurity in copper ore as a separate metallic element, which he named nickel
Nickel is a chemical element with symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive but large pieces are slow to ...
. Cronstedt is one of the founders of modern mineralogy
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the proces ...
. Cronstedt also discovered the mineral scheelite
Scheelite is a calcium tungstate mineral with the chemical formula Ca W O4. It is an important ore of tungsten (wolfram). Scheelite is originally named after Swedish chemist K. Scheele (1742-1786). Well-formed crystals are sought by collectors a ...
in 1751, which he named tungsten, meaning "heavy stone" in Swedish.
In 1754, Scottish chemist Joseph Black
Joseph Black (16 April 1728 – 6 December 1799) was a Scottish physicist and chemist, known for his discoveries of magnesium, latent heat, specific heat, and carbon dioxide. He was Professor of Anatomy and Chemistry at the University of Glas ...
isolated carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
, which he called "fixed air". In 1757, Louis Claude Cadet de Gassicourt
Louis Claude Cadet de Gassicourt (24 July 1731 – 17 October 1799) was a French chemist who synthesised the first organometalic compound.
He obtained a red liquid by the reaction of potassium acetate with arsenic trioxide. This liquid is ...
, while investigating arsenic compounds, creates Cadet's fuming liquid
Cadet's fuming liquid was a red-brown oily liquid prepared in 1760 by the French chemist Louis Claude Cadet de Gassicourt (1731-1799) by the reaction of potassium acetate with arsenic trioxide. It consisted mostly of dicacodyl (((CH3)2As)2) an ...
, later discovered to be cacodyl oxide
Cacodyl oxide is a chemical compound of the formula CH3)2Assub>2O. This organoarsenic compound is primarily of historical significance since it is sometimes considered to be the first organometallic compound synthesized in relatively pure form. ...
, considered to be the first synthetic organometallic
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and so ...
compound. In 1758, Joseph Black formulated the concept of latent heat
Latent heat (also known as latent energy or heat of transformation) is energy released or absorbed, by a body or a thermodynamic system, during a constant-temperature process — usually a first-order phase transition.
Latent heat can be understo ...
to explain the thermochemistry
Thermochemistry is the study of the heat energy which is associated with chemical reactions and/or phase changes such as melting and boiling. A reaction may release or absorb energy, and a phase change may do the same. Thermochemistry focuses on ...
of phase changes
In chemistry, thermodynamics, and other related fields, a phase transition (or phase change) is the physical process of transition between one state of a medium and another. Commonly the term is used to refer to changes among the basic states of ...
. In 1766, English chemist Henry Cavendish
Henry Cavendish ( ; 10 October 1731 – 24 February 1810) was an English natural philosopher and scientist who was an important experimental and theoretical chemist and physicist. He is noted for his discovery of hydrogen, which he termed "infl ...
isolated hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
, which he called "inflammable air". Cavendish discovered hydrogen as a colorless, odourless gas that burns and can form an explosive mixture with air, and published a paper on the production of water by burning inflammable air (that is, hydrogen) in dephlogisticated air (now known to be oxygen), the latter a constituent of atmospheric air (phlogiston theory
The phlogiston theory is a superseded scientific theory that postulated the existence of a fire-like element called phlogiston () contained within combustible bodies and released during combustion. The name comes from the Ancient Greek (''burni ...
).
In 1773, Swedish chemist Carl Wilhelm Scheele
Carl Wilhelm Scheele (, ; 9 December 1742 – 21 May 1786) was a Swedish German pharmaceutical chemist.
Scheele discovered oxygen (although Joseph Priestley published his findings first), and identified molybdenum, tungsten, barium, hydrog ...
discovered oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
, which he called "fire air", but did not immediately publish his achievement. In 1774, English chemist Joseph Priestley
Joseph Priestley (; 24 March 1733 – 6 February 1804) was an English chemist, natural philosopher, separatist theologian, grammarian, multi-subject educator, and liberal political theorist. He published over 150 works, and conducted exp ...
independently isolated oxygen in its gaseous state, calling it "dephlogisticated air", and published his work before Scheele. During his lifetime, Priestley's considerable scientific reputation rested on his invention of soda water
Carbonated water (also known as soda water, sparkling water, fizzy water, club soda, water with gas, in many places as mineral water, or especially in the United States as seltzer or seltzer water) is water containing dissolved carbon dioxide gas, ...
, his writings on electricity
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described ...
, and his discovery of several "airs" (gases), the most famous being what Priestley dubbed "dephlogisticated air" (oxygen). However, Priestley's determination to defend phlogiston theory and to reject what would become the chemical revolution
The chemical revolution, also called the ''first chemical revolution'', was the early modern reformulation of chemistry that culminated in the law of conservation of mass and the oxygen theory of combustion.
During the 19th and 20th century, thi ...
eventually left him isolated within the scientific community.
In 1781, Carl Wilhelm Scheele discovered that a new acid
In computer science, ACID ( atomicity, consistency, isolation, durability) is a set of properties of database transactions intended to guarantee data validity despite errors, power failures, and other mishaps. In the context of databases, a sequ ...
, tungstic acid
Tungstic acid refers to hydrated forms of tungsten trioxide, WO3. Both a monohydrate (WO3·H2O) and hemihydrate (WO3·1/2 H2O) are known. Molecular species akin to sulfuric acid, i.e. (HO)2WO2 are not observed.
The solid-state structure of ...
, could be made from Cronstedt's scheelite (at the time named tungsten). Scheele and Torbern Bergman
Torbern Olaf (Olof) Bergman (''KVO'') (20 March 17358 July 1784) was a Swedish chemist and mineralogist noted for his 1775 ''Dissertation on Elective Attractions'', containing the largest chemical affinity tables ever published. Bergman was the ...
suggested that it might be possible to obtain a new metal by reducing this acid.José
José is a predominantly Spanish and Portuguese form of the given name Joseph. While spelled alike, this name is pronounced differently in each language: Spanish ; Portuguese (or ).
In French, the name ''José'', pronounced , is an old vernacul ...
and Fausto Elhuyar
Fausto de Elhuyar (11 October 1755 – 6 February 1833) was a Spanish chemist, and the first to isolate tungsten with his brother Juan José Elhuyar in 1783. He was in charge, under a King of Spain commission, of organizing the School of Mines i ...
found an acid made from wolframite
Wolframite is an iron, manganese, and tungstate mineral with a chemical formula of that is the intermediate between ferberite ( rich) and hübnerite ( rich). Along with scheelite, the wolframite series are the most important tungsten ore minerals ...
that was identical to tungstic acid. Later that year, in Spain, the brothers succeeded in isolating the metal now known as tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isolat ...
by reduction of this acid with charcoal
Charcoal is a lightweight black carbon residue produced by strongly heating wood (or other animal and plant materials) in minimal oxygen to remove all water and volatile constituents. In the traditional version of this pyrolysis process, cal ...
, and they are credited with the discovery of the element.
Volta and the Voltaic pile
Italian physicist Alessandro Volta
Alessandro Giuseppe Antonio Anastasio Volta (, ; 18 February 1745 – 5 March 1827) was an Italian physicist, chemist and lay Catholic who was a pioneer of electricity and power who is credited as the inventor of the electric battery and the ...
constructed a device for accumulating a large charge by a series of inductions and groundings. He investigated the 1780s discovery " animal electricity" by Luigi Galvani
Luigi Galvani (, also ; ; la, Aloysius Galvanus; 9 September 1737 – 4 December 1798) was an Italian physician, physicist, biologist and philosopher, who studied animal electricity. In 1780, he discovered that the muscles of dead frogs' legs ...
, and found that the electric current
An electric current is a stream of charged particles, such as electrons or ions, moving through an electrical conductor or space. It is measured as the net rate of flow of electric charge through a surface or into a control volume. The moving pa ...
was generated from the contact of dissimilar metals, and that the frog leg was only acting as a detector. Volta demonstrated in 1794 that when two metals and brine-soaked cloth or cardboard are arranged in a circuit they produce an electric
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described by ...
current.
In 1800, Volta stacked several pairs of alternating copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
(or silver
Silver is a chemical element with the Symbol (chemistry), symbol Ag (from the Latin ', derived from the Proto-Indo-European wikt:Reconstruction:Proto-Indo-European/h₂erǵ-, ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, whi ...
) and zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
discs (electrode
An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit (e.g. a semiconductor, an electrolyte, a vacuum or air). Electrodes are essential parts of batteries that can consist of a variety of materials de ...
s) separated by cloth or cardboard soaked in brine
Brine is a high-concentration solution of salt (NaCl) in water (H2O). In diverse contexts, ''brine'' may refer to the salt solutions ranging from about 3.5% (a typical concentration of seawater, on the lower end of that of solutions used for br ...
(electrolyte
An electrolyte is a medium containing ions that is electrically conducting through the movement of those ions, but not conducting electrons. This includes most soluble salts, acids, and bases dissolved in a polar solvent, such as water. Upon dis ...
) to increase the electrolyte conductivity.current
Currents, Current or The Current may refer to:
Science and technology
* Current (fluid), the flow of a liquid or a gas
** Air current, a flow of air
** Ocean current, a current in the ocean
*** Rip current, a kind of water current
** Current (stre ...
flowed through this voltaic pile
upright=1.2, Schematic diagram of a copper–zinc voltaic pile. The copper and zinc discs were separated by cardboard or felt spacers soaked in salt water (the electrolyte). Volta's original piles contained an additional zinc disk at the bottom, ...
and the connecting wire. Thus, Volta is credited with constructing the first electrical battery
An electric battery is a source of electric power consisting of one or more electrochemical cells with external connections for powering electrical devices.
When a battery is supplying power, its positive terminal is the cathode and its negati ...
to produce electricity
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described ...
.
Thus, Volta is considered to be the founder of the discipline of electrochemistry
Electrochemistry is the branch of physical chemistry concerned with the relationship between electrical potential difference, as a measurable and quantitative phenomenon, and identifiable chemical change, with the potential difference as an outco ...
. A Galvanic cell
A galvanic cell or voltaic cell, named after the scientists Luigi Galvani and Alessandro Volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous Oxidation-Reduction reactions. A common apparatus ...
(or voltaic cell) is an electrochemical cell
An electrochemical cell is a device capable of either generating electrical energy from chemical reactions or using electrical energy to cause chemical reactions. The electrochemical cells which generate an electric current are called voltaic o ...
that derives electrical energy from a spontaneous redox
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate (chemistry), substrate change. Oxidation is the loss of Electron, electrons or an increase in the oxidation state, while reduction ...
reaction taking place within the cell. It generally consists of two different metals connected by a salt bridge
In electrochemistry, a salt bridge or ion bridge is a laboratory device used to connect the oxidation and reduction half-cells of a galvanic cell (voltaic cell), a type of electrochemical cell. It maintains electrical neutrality within the in ...
, or individual half-cells separated by a porous membrane.
Antoine-Laurent de Lavoisier
 Antoine-Laurent de Lavoisier demonstrated with careful measurements that transmutation of water to earth was not possible, but that the sediment observed from boiling water came from the container. He burnt phosphorus and sulfur in air, and proved that the products weighed more than the original samples, with the mass gained being lost from the air. Thus, in 1789, he established the Law of
Antoine-Laurent de Lavoisier demonstrated with careful measurements that transmutation of water to earth was not possible, but that the sediment observed from boiling water came from the container. He burnt phosphorus and sulfur in air, and proved that the products weighed more than the original samples, with the mass gained being lost from the air. Thus, in 1789, he established the Law of Conservation of Mass
In physics and chemistry, the law of conservation of mass or principle of mass conservation states that for any system closed to all transfers of matter and energy, the mass of the system must remain constant over time, as the system's mass can ...
, which is also called "Lavoisier's Law."
Repeating the experiments of Priestley, he demonstrated that air is composed of two parts, one of which combines with metals to form calx
Calx is a substance formed from an ore or mineral that has been heated.
Calx, especially of a metal, is now known as an oxide. According to the obsolete phlogiston theory, the calx was the true elemental substance, having lost its phlogiston in t ...
es. In ''Considérations Générales sur la Nature des Acides'' (1778), he demonstrated that the "air" responsible for combustion was also the source of acidity. The next year, he named this portion oxygen (Greek for acid-former), and the other azote (Greek for no life). Because of his more thorough characterization of it as an element, Lavoisier thus has a claim to the discovery of oxygen along with Priestley and Scheele. He also discovered that the "inflammable air" discovered by Cavendish – which he termed hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
(Greek for water-former) – combined with oxygen to produce a dew, as Priestley had reported, which appeared to be water. In ''Reflexions sur le Phlogistique'' (1783), Lavoisier showed the phlogiston theory
The phlogiston theory is a superseded scientific theory that postulated the existence of a fire-like element called phlogiston () contained within combustible bodies and released during combustion. The name comes from the Ancient Greek (''burni ...
of combustion to be inconsistent. Mikhail Lomonosov
Mikhail Vasilyevich Lomonosov (; russian: Михаил (Михайло) Васильевич Ломоносов, p=mʲɪxɐˈil vɐˈsʲilʲjɪvʲɪtɕ , a=Ru-Mikhail Vasilyevich Lomonosov.ogg; – ) was a Russian Empire, Russian polymath, s ...
independently established a tradition of chemistry in Russia in the 18th century; he also rejected the phlogiston theory, and anticipated the kinetic theory of gases
Kinetic (Ancient Greek: κίνησις “kinesis”, movement or to move) may refer to:
* Kinetic theory, describing a gas as particles in random motion
* Kinetic energy, the energy of an object that it possesses due to its motion
Art and enter ...
. Lomonosov regarded heat as a form of motion, and stated the idea of conservation of matter.
Lavoisier worked with Claude Louis Berthollet
Claude Louis Berthollet (, 9 December 1748 – 6 November 1822) was a Savoyard-French chemist who became vice president of the French Senate in 1804. He is known for his scientific contributions to theory of chemical equilibria via the mecha ...
and others to devise a system of chemical nomenclature
A chemical nomenclature is a set of rules to generate systematic names for chemical compounds. The nomenclature used most frequently worldwide is the one created and developed by the International Union of Pure and Applied Chemistry (IUPAC).
The ...
, which serves as the basis of the modern system of naming chemical compounds. In his ''Methods of Chemical Nomenclature'' (1787), Lavoisier invented the system of naming and classification still largely in use today, including names such as sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid ( Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with the molecular formu ...
, sulfate
The sulfate or sulphate ion is a polyatomic anion with the empirical formula . Salts, acid derivatives, and peroxides of sulfate are widely used in industry. Sulfates occur widely in everyday life. Sulfates are salts of sulfuric acid and many ar ...
s, and sulfite
Sulfites or sulphites are compounds that contain the sulfite ion (or the sulfate(IV) ion, from its correct systematic name), . The sulfite ion is the conjugate base of bisulfite. Although its acid ( sulfurous acid) is elusive, its salts are wide ...
s. In 1785, Berthollet was the first to introduce the use of chlorine gas as a commercial bleach. In the same year he first determined the elemental composition of the gas ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
. Berthollet first produced a modern bleaching liquid in 1789 by passing chlorine gas through a solution of sodium carbonate
Sodium carbonate, , (also known as washing soda, soda ash and soda crystals) is the inorganic compound with the formula Na2CO3 and its various hydrates. All forms are white, odourless, water-soluble salts that yield moderately alkaline solutions ...
– the result was a weak solution of sodium hypochlorite
Sodium hypochlorite (commonly known in a dilute solution as bleach) is an Inorganic chemistry, inorganic chemical compound with the chemical formula, formula NaOCl (or NaClO), comprising a sodium cation () and a hypochlorite anion (or ). It may ...
. Another strong chlorine oxidant and bleach which he investigated and was the first to produce, potassium chlorate
Potassium chlorate is a compound containing potassium, chlorine and oxygen, with the molecular formula KClO3. In its pure form, it is a white crystalline substance. After sodium chlorate, it is the second most common chlorate in industrial use. It ...
(KClO3), is known as Berthollet's Salt. Berthollet is also known for his scientific contributions to the theory of chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both the reactants and products are present in concentrations which have no further tendency to change with time, so that there is no observable change in the properties of the sy ...
via the mechanism of reversible reaction
A reversible reaction is a reaction in which the conversion of reactants to products and the conversion of products to reactants occur simultaneously.
: \mathit aA + \mathit bB \mathit cC + \mathit dD
A and B can react to form C and D or, in the ...
s.
Lavoisier's ''Traité Élémentaire de Chimie
''Traité élémentaire de chimie'' (''Elementary Treatise on Chemistry'') is a textbook written by Antoine Lavoisier published in 1789 and translated into English by Robert Kerr in 1790 under the title ''Elements of Chemistry in a New Systemati ...
'' (Elementary Treatise of Chemistry, 1789) was the first modern chemical textbook, and presented a unified view of new theories of chemistry, contained a clear statement of the Law of Conservation of Mass, and denied the existence of phlogiston. In addition, it contained a list of elements, or substances that could not be broken down further, which included oxygen, nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
, hydrogen, phosphorus
Phosphorus is a chemical element with the symbol P and atomic number 15. Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Ear ...
, mercury
Mercury commonly refers to:
* Mercury (planet), the nearest planet to the Sun
* Mercury (element), a metallic chemical element with the symbol Hg
* Mercury (mythology), a Roman god
Mercury or The Mercury may also refer to:
Companies
* Merc ...
, zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
, and sulfur
Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula ...
. His list, however, also included light and caloric, which he believed to be material substances. In the work, Lavoisier underscored the observational basis of his chemistry, stating "I have tried...to arrive at the truth by linking up facts; to suppress as much as possible the use of reasoning, which is often an unreliable instrument which deceives us, in order to follow as much as possible the torch of observation and of experiment." Nevertheless, he believed that the real existence of atoms was philosophically impossible. Lavoisier demonstrated that organisms disassemble and reconstitute atmospheric air in the same manner as a burning body.
With Pierre-Simon Laplace
Pierre-Simon, marquis de Laplace (; ; 23 March 1749 – 5 March 1827) was a French scholar and polymath whose work was important to the development of engineering, mathematics, statistics, physics, astronomy, and philosophy. He summarized ...
, Lavoisier used a calorimeter
A calorimeter is an object used for calorimetry, or the process of measuring the heat of chemical reactions or physical changes as well as heat capacity. Differential scanning calorimeters, isothermal micro calorimeters, titration calorimete ...
to estimate the heat evolved per unit of carbon dioxide produced. They found the same ratio for a flame and animals, indicating that animals produced energy by a type of combustion. Lavoisier believed in the radical theory Radical theory is an obsolete scientific theory in chemistry describing the structure of organic compounds. The theory was pioneered by Justus von Liebig, Friedrich Wöhler and Auguste Laurent around 1830 and is not related to the modern understand ...
, which stated that radicals, which function as a single group in a chemical reaction, would combine with oxygen in reactions. He believed all acids contained oxygen. He also discovered that diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
is a crystalline form of carbon.
Although many of Lavoisier's partners were influential for the advancement of chemistry as a scientific discipline, his wife Marie-Anne Lavoisier was arguably the most influential of them all. Upon their marriage, Mme. Lavoisier began to study chemistry, English, and drawing in order to help her husband in his work either by translating papers into English, a language which Lavoisier did not know, or by keeping records and drawing the various apparatuses that Lavoisier used in his labs.Richard Kirwan
Richard Kirwan, LL.D, FRS, FRSE MRIA (1 August 1733 – 22 June 1812) was an Irish geologist and chemist. He was one of the last supporters of the theory of phlogiston.
Kirwan was active in the fields of chemistry, meteorology, and geol ...
. While translating, she stumbled upon and corrected multiple errors. When she presented her translation, along with her notes, to Lavoisier, her contributions led to Lavoisier's refutation of the theory of phlogiston.
Lavoisier made many fundamental contributions to the science of chemistry. Following his work, chemistry acquired a strict, quantitative nature, allowing reliable predictions to be made. The revolution in chemistry which he brought about was a result of a conscious effort to fit all experiments into the framework of a single theory. He established the consistent use of chemical balance, used oxygen to overthrow the phlogiston theory, and developed a new system of chemical nomenclature. Further potential contributions were cut short when Lavoisier was beheaded during the French Revolution
The French Revolution ( ) was a period of radical political and societal change in France that began with the Estates General of 1789 and ended with the formation of the French Consulate in November 1799. Many of its ideas are considere ...
.
19th century
In 1802, French American chemist and industrialist Éleuthère Irénée du Pont, who learned manufacture of gunpowder and explosives under Antoine Lavoisier, founded a gunpowder manufacturer in Delaware known as DuPont, E. I. du Pont de Nemours and Company. The French Revolution
The French Revolution ( ) was a period of radical political and societal change in France that began with the Estates General of 1789 and ended with the formation of the French Consulate in November 1799. Many of its ideas are considere ...
forced his family to move to the United States where du Pont started a gunpowder mill on the Brandywine River in Delaware. Wanting to make the best powder possible, du Pont was vigilant about the quality of the materials he used. For 32 years, du Pont served as president of E. I. du Pont de Nemours and Company, which eventually grew into one of the largest and most successful companies in America.
Throughout the 19th century, chemistry was divided between those who followed the atomic theory of John Dalton and those who did not, such as Wilhelm Ostwald and Ernst Mach.[
] Although such proponents of the atomic theory as Amedeo Avogadro and Ludwig Boltzmann made great advances in explaining the behavior of gas
Gas is one of the four fundamental states of matter (the others being solid, liquid, and plasma).
A pure gas may be made up of individual atoms (e.g. a noble gas like neon), elemental molecules made from one type of atom (e.g. oxygen), or ...
es, this dispute was not finally settled until Jean Perrin's experimental investigation of Albert Einstein, Einstein's atomic explanation of Brownian motion in the first decade of the 20th century.
Vladimir Markovnikov
Vladimir Markovnikov born in 1838, was a Russian Scientist who proceeded most of his work at Kazan University in Russia.
John Dalton
 In 1803, English meteorologist and chemist John Dalton proposed Dalton's law, which describes the relationship between the components in a mixture of gases and the relative pressure each contributes to that of the overall mixture.
In 1803, English meteorologist and chemist John Dalton proposed Dalton's law, which describes the relationship between the components in a mixture of gases and the relative pressure each contributes to that of the overall mixture.
Jöns Jacob Berzelius
 A Swedish chemist and disciple of Dalton, Jöns Jacob Berzelius embarked on a systematic program to try to make accurate and precise quantitative measurements and to ensure the purity of chemicals. Along with Lavoisier, Boyle, and Dalton, Berzelius is known as the father of modern chemistry. In 1828 he compiled a table of relative atomic weights, where
A Swedish chemist and disciple of Dalton, Jöns Jacob Berzelius embarked on a systematic program to try to make accurate and precise quantitative measurements and to ensure the purity of chemicals. Along with Lavoisier, Boyle, and Dalton, Berzelius is known as the father of modern chemistry. In 1828 he compiled a table of relative atomic weights, where oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
was used as a standard, with its weight set at 100, and which included all of the elements known at the time. This work provided evidence in favor of Dalton's atomic theory – that inorganic chemical compounds are composed of atoms combined in Whole number rule, whole number amounts. He determined the exact elementary constituents of a large number of compounds; the results strongly supported Proust's Law of Definite Proportions. In discovering that atomic weights are not integer multiples of the weight of hydrogen, Berzelius also disproved Prout's hypothesis that elements are built up from atoms of hydrogen.
Motivated by his extensive atomic weight determinations and in a desire to aid his experiments, he introduced the classical system of chemical symbols and notation with his 1808 publication ''Lärbok i Kemien'', in which elements are abbreviated to one or two letters to make a distinct symbol from their Latin name. This system of chemical notation—in which the elements were given simple written labels, such as O for oxygen, or Fe for iron, with proportions denoted by numbers—is the same basic system used today. The only difference is that instead of the subscript number used today (e.g., H2O), Berzelius used a superscript (H2O). Berzelius is credited with identifying the chemical elements silicon, selenium, thorium, and cerium. Students working in Berzelius's laboratory also discovered lithium and vanadium.
Berzelius developed the radical theory Radical theory is an obsolete scientific theory in chemistry describing the structure of organic compounds. The theory was pioneered by Justus von Liebig, Friedrich Wöhler and Auguste Laurent around 1830 and is not related to the modern understand ...
of chemical combination, which holds that reactions occur as stable groups of atoms called Radical (chemistry), radicals are exchanged between molecules. He believed that salts are compounds formed of acids and Base (chemistry), bases, and discovered that the anions in acids were attracted to a positive electrode (the anode), whereas the cations in a base were attracted to a negative electrode (the cathode). Berzelius did not believe in the Vitalism Theory, but instead in a regulative force which produced organization of tissues in an organism. Berzelius is also credited with originating the chemical terms "catalysis", "polymer", "isomer", and "allotrope", although his original definitions differ dramatically from modern usage. For example, he coined the term "polymer" in 1833 to describe organic compounds which shared identical empirical formulas but which differed in overall molecular weight, the larger of the compounds being described as "polymers" of the smallest. By this long-superseded, pre-structural definition, glucose (C6H12O6) was viewed as a polymer of formaldehyde (CH2O).
New elements and gas laws
 English chemist Humphry Davy was a pioneer in the field of electrolysis, using Alessandro Volta's voltaic pile to split up common compounds and thus isolate a series of new elements. He went on to electrolyse molten salts and discovered several new metals, especially sodium and potassium, highly reactive elements known as the alkali metals. Potassium, the first metal that was isolated by electrolysis, was discovered in 1807 by Davy, who derived it from potassium hydroxide, caustic potash (KOH). Before the 19th century, no distinction was made between potassium and sodium. Sodium was first isolated by Davy in the same year by passing an electric current through molten sodium hydroxide (NaOH). When Davy heard that Berzelius and Pontin prepared calcium amalgam by electrolyzing lime in mercury, he tried it himself. Davy was successful, and discovered calcium in 1808 by electrolyzing a mixture of Lime (material), lime and mercuric oxide.
English chemist Humphry Davy was a pioneer in the field of electrolysis, using Alessandro Volta's voltaic pile to split up common compounds and thus isolate a series of new elements. He went on to electrolyse molten salts and discovered several new metals, especially sodium and potassium, highly reactive elements known as the alkali metals. Potassium, the first metal that was isolated by electrolysis, was discovered in 1807 by Davy, who derived it from potassium hydroxide, caustic potash (KOH). Before the 19th century, no distinction was made between potassium and sodium. Sodium was first isolated by Davy in the same year by passing an electric current through molten sodium hydroxide (NaOH). When Davy heard that Berzelius and Pontin prepared calcium amalgam by electrolyzing lime in mercury, he tried it himself. Davy was successful, and discovered calcium in 1808 by electrolyzing a mixture of Lime (material), lime and mercuric oxide.Carl Wilhelm Scheele
Carl Wilhelm Scheele (, ; 9 December 1742 – 21 May 1786) was a Swedish German pharmaceutical chemist.
Scheele discovered oxygen (although Joseph Priestley published his findings first), and identified molybdenum, tungsten, barium, hydrog ...
, who called it ''"dephlogisticated marine acid"'' (see phlogiston theory
The phlogiston theory is a superseded scientific theory that postulated the existence of a fire-like element called phlogiston () contained within combustible bodies and released during combustion. The name comes from the Ancient Greek (''burni ...
) and mistakenly thought it contained oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
. Scheele observed several properties of chlorine gas, such as its bleaching effect on litmus, its deadly effect on insects, its yellow-green colour, and the similarity of its smell to that of aqua regia. However, Scheele was unable to publish his findings at the time. In 1810, chlorine was given its current name by Humphry Davy (derived from the Greek word for green), who insisted that chlorine was in fact an Chemical element, element. He also showed that oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
could not be obtained from the substance known as Chlorine#History, oxymuriatic acid (HCl solution). This discovery overturned Antoine Lavoisier, Lavoisier's definition of acids as compounds of oxygen. Davy was a popular lecturer and able experimenter.
 French chemist Joseph Louis Gay-Lussac shared the interest of Lavoisier and others in the quantitative study of the properties of gases. From his first major program of research in 1801–1802, he concluded that equal volumes of all gases expand equally with the same increase in temperature: this conclusion is usually called "Charles's law", as Gay-Lussac gave credit to Jacques Charles, who had arrived at nearly the same conclusion in the 1780s but had not published it.
French chemist Joseph Louis Gay-Lussac shared the interest of Lavoisier and others in the quantitative study of the properties of gases. From his first major program of research in 1801–1802, he concluded that equal volumes of all gases expand equally with the same increase in temperature: this conclusion is usually called "Charles's law", as Gay-Lussac gave credit to Jacques Charles, who had arrived at nearly the same conclusion in the 1780s but had not published it.
English translation (extract).
br />
On page 157, Gay-Lussac mentions the unpublished findings of Charles: "''Avant d'aller plus loin, je dois prévenir que quoique j'eusse reconnu un grand nombre de fois que les gaz oxigène, azote, hydrogène et acide carbonique, et l'air atmosphérique se dilatent également depuis 0° jusqu'a 80°, le cit. Charles avait remarqué depuis 15 ans la même propriété dans ces gaz ; mais n'avant jamais publié ses résultats, c'est par le plus grand hasard que je les ai connus''." (Before going further, I should inform [you] that although I had recognized many times that the gases oxygen, nitrogen, hydrogen, and carbonic acid [i.e., carbon dioxide], and atmospheric air also expand from 0° to 80°, citizen Charles had noticed 15 years ago the same property in these gases; but having never published his results, it is by the merest chance that I knew of them.)[ In French, seaweed that had been washed onto the shore was called "varec", "varech", or "vareck", whence the English word "wrack". Later, "varec" also referred to the ashes of such seaweed: The ashes were used as a source of iodine and salts of sodium and potassium.] Courtois gave samples to his friends, Charles Bernard Desormes (1777–1862) and Nicolas Clément (1779–1841), to continue research. He also gave some of the substance to Gay-Lussac and to physicist André-Marie Ampère. On December 6, 1813, Gay-Lussac announced that the new substance was either an element or a compound of oxygen.[David Knight, ‘Davy, Sir Humphry, baronet (1778–1829)’, Oxford Dictionary of National Biography, Oxford University Press, 200]
accessed 6 April 2008
/ref>
 After Dalton published his atomic theory in 1808, certain of his central ideas were soon adopted by most chemists. However, uncertainty persisted for half a century about how atomic theory was to be configured and applied to concrete situations; chemists in different countries developed several different incompatible atomistic systems. A paper that suggested a way out of this difficult situation was published as early as 1811 by the Italian physicist Amedeo Avogadro (1776–1856), who hypothesized that equal volumes of gases at the same temperature and
After Dalton published his atomic theory in 1808, certain of his central ideas were soon adopted by most chemists. However, uncertainty persisted for half a century about how atomic theory was to be configured and applied to concrete situations; chemists in different countries developed several different incompatible atomistic systems. A paper that suggested a way out of this difficult situation was published as early as 1811 by the Italian physicist Amedeo Avogadro (1776–1856), who hypothesized that equal volumes of gases at the same temperature and pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and e ...
contain equal numbers of molecules, from which it followed that relative molecular weights of any two gases are the same as the ratio of the densities of the two gases under the same conditions of temperature and pressure. Avogadro also reasoned that simple gases were not formed of solitary atoms but were instead compound molecules of two or more atoms. Thus Avogadro was able to overcome the difficulty that Dalton and others had encountered when Gay-Lussac reported that above 100 °C the volume of water vapor was twice the volume of the oxygen used to form it. According to Avogadro, the molecule of oxygen had split into two atoms in the course of forming water vapor.
Avogadro's hypothesis was neglected for half a century after it was first published. Many reasons for this neglect have been cited, including some theoretical problems, such as Jöns Jacob Berzelius's "dualism", which asserted that compounds are held together by the attraction of positive and negative electrical charges, making it inconceivable that a molecule composed of two electrically similar atoms—as in oxygen—could exist. An additional barrier to acceptance was the fact that many chemists were reluctant to adopt physical methods (such as vapour-density determinations) to solve their problems. By mid-century, however, some leading figures had begun to view the chaotic multiplicity of competing systems of atomic weights and molecular formulas as intolerable. Moreover, purely chemical evidence began to mount that suggested Avogadro's approach might be right after all. During the 1850s, younger chemists, such as Alexander William Williamson, Alexander Williamson in England, Charles Frédéric Gerhardt, Charles Gerhardt and Charles-Adolphe Wurtz in France, and August Kekulé in Germany, began to advocate reforming theoretical chemistry to make it consistent with Avogadrian theory.
Wöhler and the vitalism debate
 In 1825, Friedrich Wöhler and Justus von Liebig performed the first confirmed discovery and explanation of isomers, earlier named by Berzelius. Working with cyanic acid and fulminic acid, they correctly deduced that isomerism was caused by differing arrangements of atoms within a molecular structure. In 1827, William Prout classified biomolecules into their modern groupings: carbohydrates, proteins and lipids. After the nature of combustion was settled, a dispute about vitalism and the essential distinction between organic and inorganic substances began. The vitalism question was revolutionized in 1828 when Friedrich Wöhler synthesized urea, thereby establishing that organic compounds could be produced from inorganic starting materials and disproving the theory of vitalism.
This opened a new research field in chemistry, and by the end of the 19th century, scientists were able to synthesize hundreds of organic compounds. The most important among them are mauve, magenta, and other synthetic dyes, as well as the widely used drug aspirin. The discovery of the artificial synthesis of urea contributed greatly to the theory of isomerism, as the empirical chemical formulas for urea and ammonium cyanate are identical (see Wöhler synthesis). In 1832, Friedrich Wöhler and Justus von Liebig discovered and explained functional groups and radical (chemistry), radicals in relation to organic chemistry, as well as first synthesizing benzaldehyde. Liebig, a German chemist, made major contributions to agriculture, agricultural and biochemistry, biological chemistry, and worked on the organization of organic chemistry. Liebig is considered the "father of the fertilizer industry" for his discovery of
In 1825, Friedrich Wöhler and Justus von Liebig performed the first confirmed discovery and explanation of isomers, earlier named by Berzelius. Working with cyanic acid and fulminic acid, they correctly deduced that isomerism was caused by differing arrangements of atoms within a molecular structure. In 1827, William Prout classified biomolecules into their modern groupings: carbohydrates, proteins and lipids. After the nature of combustion was settled, a dispute about vitalism and the essential distinction between organic and inorganic substances began. The vitalism question was revolutionized in 1828 when Friedrich Wöhler synthesized urea, thereby establishing that organic compounds could be produced from inorganic starting materials and disproving the theory of vitalism.
This opened a new research field in chemistry, and by the end of the 19th century, scientists were able to synthesize hundreds of organic compounds. The most important among them are mauve, magenta, and other synthetic dyes, as well as the widely used drug aspirin. The discovery of the artificial synthesis of urea contributed greatly to the theory of isomerism, as the empirical chemical formulas for urea and ammonium cyanate are identical (see Wöhler synthesis). In 1832, Friedrich Wöhler and Justus von Liebig discovered and explained functional groups and radical (chemistry), radicals in relation to organic chemistry, as well as first synthesizing benzaldehyde. Liebig, a German chemist, made major contributions to agriculture, agricultural and biochemistry, biological chemistry, and worked on the organization of organic chemistry. Liebig is considered the "father of the fertilizer industry" for his discovery of nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
as an essential plant nutrient, and his formulation of the Law of the Minimum which described the effect of individual nutrients on crops.
Mid-1800s
In 1840, Germain Hess proposed Hess's law, an early statement of the law of conservation of energy, which establishes that energy changes in a chemical process depend only on the states of the starting and product materials and not on the specific pathway taken between the two states. In 1847, Adolph Wilhelm Hermann Kolbe, Hermann Kolbe obtained acetic acid from completely inorganic sources, further disproving vitalism. In 1848, William Thomson, 1st Baron Kelvin (commonly known as Lord Kelvin) established the concept of absolute zero, the temperature at which all molecular motion ceases. In 1849, Louis Pasteur discovered that the racemic form of tartaric acid is a mixture of the levorotatory and dextrotatory forms, thus clarifying the nature of optical rotation and advancing the field of stereochemistry. In 1852, August Beer proposed Beer's law, which explains the relationship between the composition of a mixture and the amount of light it will absorb. Based partly on earlier work by Pierre Bouguer and Johann Heinrich Lambert, it established the analytical chemistry, analytical technique known as spectrophotometry. In 1855, Benjamin Silliman, Jr. pioneered methods of petroleum cracking, which made the entire modern petrochemical industry possible.
 Avogadro's hypothesis began to gain broad appeal among chemists only after his compatriot and fellow scientist Stanislao Cannizzaro demonstrated its value in 1858, two years after Avogadro's death. Cannizzaro's chemical interests had originally centered on natural products and on reactions of aromatic compounds; in 1853 he discovered that when benzaldehyde is treated with concentrated base, both benzoic acid and benzyl alcohol are produced—a phenomenon known today as the Cannizzaro reaction. In his 1858 pamphlet, Cannizzaro showed that a complete return to the ideas of Avogadro could be used to construct a consistent and robust theoretical structure that fit nearly all of the available empirical evidence. For instance, he pointed to evidence that suggested that not all elementary gases consist of two atoms per molecule—some were monatomic, most were diatomic, and a few were even more complex.
Another point of contention had been the formulas for compounds of the alkali metals (such as sodium) and the alkaline earth metals (such as calcium), which, in view of their striking chemical analogies, most chemists had wanted to assign to the same formula type. Cannizzaro argued that placing these metals in different categories had the beneficial result of eliminating certain anomalies when using their physical properties to deduce atomic weights. Unfortunately, Cannizzaro's pamphlet was published initially only in Italian and had little immediate impact. The real breakthrough came with an Karlsruhe Congress, international chemical congress held in the German town of Karlsruhe in September 1860, at which most of the leading European chemists were present. The Karlsruhe Congress had been arranged by Kekulé, Wurtz, and a few others who shared Cannizzaro's sense of the direction chemistry should go. Speaking in French (as everyone there did), Cannizzaro's eloquence and logic made an indelible impression on the assembled body. Moreover, his friend Angelo Pavesi distributed Cannizzaro's pamphlet to attendees at the end of the meeting; more than one chemist later wrote of the decisive impression the reading of this document provided. For instance, Julius Lothar Meyer, Lothar Meyer later wrote that on reading Cannizzaro's paper, "The scales seemed to fall from my eyes." Cannizzaro thus played a crucial role in winning the battle for reform. The system advocated by him, and soon thereafter adopted by most leading chemists, is substantially identical to what is still used today.
Avogadro's hypothesis began to gain broad appeal among chemists only after his compatriot and fellow scientist Stanislao Cannizzaro demonstrated its value in 1858, two years after Avogadro's death. Cannizzaro's chemical interests had originally centered on natural products and on reactions of aromatic compounds; in 1853 he discovered that when benzaldehyde is treated with concentrated base, both benzoic acid and benzyl alcohol are produced—a phenomenon known today as the Cannizzaro reaction. In his 1858 pamphlet, Cannizzaro showed that a complete return to the ideas of Avogadro could be used to construct a consistent and robust theoretical structure that fit nearly all of the available empirical evidence. For instance, he pointed to evidence that suggested that not all elementary gases consist of two atoms per molecule—some were monatomic, most were diatomic, and a few were even more complex.
Another point of contention had been the formulas for compounds of the alkali metals (such as sodium) and the alkaline earth metals (such as calcium), which, in view of their striking chemical analogies, most chemists had wanted to assign to the same formula type. Cannizzaro argued that placing these metals in different categories had the beneficial result of eliminating certain anomalies when using their physical properties to deduce atomic weights. Unfortunately, Cannizzaro's pamphlet was published initially only in Italian and had little immediate impact. The real breakthrough came with an Karlsruhe Congress, international chemical congress held in the German town of Karlsruhe in September 1860, at which most of the leading European chemists were present. The Karlsruhe Congress had been arranged by Kekulé, Wurtz, and a few others who shared Cannizzaro's sense of the direction chemistry should go. Speaking in French (as everyone there did), Cannizzaro's eloquence and logic made an indelible impression on the assembled body. Moreover, his friend Angelo Pavesi distributed Cannizzaro's pamphlet to attendees at the end of the meeting; more than one chemist later wrote of the decisive impression the reading of this document provided. For instance, Julius Lothar Meyer, Lothar Meyer later wrote that on reading Cannizzaro's paper, "The scales seemed to fall from my eyes." Cannizzaro thus played a crucial role in winning the battle for reform. The system advocated by him, and soon thereafter adopted by most leading chemists, is substantially identical to what is still used today.
Perkin, Crookes, and Nobel
In 1856, Sir William Henry Perkin, age 18, given a challenge by his professor, August Wilhelm von Hofmann, sought to synthesize quinine total synthesis, quinine, the anti-malaria drug, from coal tar. In one attempt, Perkin oxidation, oxidized aniline using potassium dichromate, whose toluidine impurities reacted with the aniline and yielded a black solid—suggesting a "failed" organic synthesis. Cleaning the flask with alcohol, Perkin noticed purple portions of the solution: a byproduct of the attempt was the first synthetic dye, known as mauveine or Perkin's mauve. Perkin's discovery is the foundation of the dye synthesis industry, one of the earliest successful chemical industries.
German chemist August Kekulé, August Kekulé von Stradonitz's most important single contribution was his structural theory of organic composition, outlined in two articles published in 1857 and 1858 and treated in great detail in the pages of his extraordinarily popular ''Lehrbuch der organischen Chemie'' ("Textbook of Organic Chemistry"), the first installment of which appeared in 1859 and gradually extended to four volumes. Kekulé argued that tetravalent carbon atoms – that is, carbon forming exactly four chemical bonds – could link together to form what he called a "carbon chain" or a "carbon skeleton," to which other atoms with other valences (such as hydrogen, oxygen, nitrogen, and chlorine) could join. He was convinced that it was possible for the chemist to specify this detailed molecular architecture for at least the simpler organic compounds known in his day. Kekulé was not the only chemist to make such claims in this era. The Scottish chemist Archibald Scott Couper published a substantially similar theory nearly simultaneously, and the Russian chemist Aleksandr Butlerov did much to clarify and expand structure theory. However, it was predominantly Kekulé's ideas that prevailed in the chemical community.
 British chemist and physicist William Crookes is noted for his cathode ray studies, fundamental in the development of atomic physics. His researches on electrical discharges through a rarefied gas led him to observe the dark space around the cathode, now called the Crookes dark space. He demonstrated that cathode rays travel in straight lines and produce phosphorescence and heat when they strike certain materials. A pioneer of vacuum tubes, Crookes invented the Crookes tube – an early experimental discharge tube, with partial vacuum with which he studied the behavior of cathode rays. With the introduction of Spectroscopy, spectrum analysis by Robert Bunsen and Gustav Kirchhoff (1859–1860), Crookes applied the new technique to the study of selenium compounds. Bunsen and Kirchhoff had previously used spectroscopy as a means of chemical analysis to discover caesium and rubidium. In 1861, Crookes used this process to discover thallium in some seleniferous deposits. He continued work on that new element, isolated it, studied its properties, and in 1873 determined its atomic weight. During his studies of thallium, Crookes discovered the principle of the Crookes radiometer, a device that converts light radiation into rotary motion. The principle of this radiometer has found numerous applications in the development of sensitive measuring instruments.
In 1862, Alexander Parkes exhibited Parkesine, one of the earliest synthetic polymers, at the International Exhibition in London. This discovery formed the foundation of the modern plastics industry. In 1864, Cato Maximilian Guldberg and Peter Waage, building on Claude Louis Berthollet's ideas, proposed the law of mass action. In 1865, Johann Josef Loschmidt determined the number of molecules in a mole (unit), mole, later named Avogadro constant, Avogadro's number.
In 1865, August Kekulé, based partially on the work of Loschmidt and others, established the structure of benzene as a six carbon ring with alternating single and double bonds. Kekulé's novel proposal for benzene's cyclic structure was much contested but was never replaced by a superior theory. This theory provided the scientific basis for the dramatic expansion of the German chemical industry in the last third of the 19th century. Kekulé is also famous for having clarified the nature of aromatic compounds, which are compounds based on the benzene molecule. In 1865, Adolf von Baeyer began work on indigo dye, a milestone in modern industrial organic chemistry which revolutionized the dye industry.
Swedish chemist and inventor Alfred Nobel found that when nitroglycerin was incorporated in an absorbent inert substance like ''kieselguhr'' (diatomaceous earth) it became safer and more convenient to handle, and this mixture he patented in 1867 as dynamite. Nobel later on combined nitroglycerin with various nitrocellulose compounds, similar to collodion, but settled on a more efficient recipe combining another nitrate explosive, and obtained a transparent, jelly-like substance, which was a more powerful explosive than dynamite. Gelignite, or blasting gelatin, as it was named, was patented in 1876; and was followed by a host of similar combinations, modified by the addition of potassium nitrate and various other substances.
British chemist and physicist William Crookes is noted for his cathode ray studies, fundamental in the development of atomic physics. His researches on electrical discharges through a rarefied gas led him to observe the dark space around the cathode, now called the Crookes dark space. He demonstrated that cathode rays travel in straight lines and produce phosphorescence and heat when they strike certain materials. A pioneer of vacuum tubes, Crookes invented the Crookes tube – an early experimental discharge tube, with partial vacuum with which he studied the behavior of cathode rays. With the introduction of Spectroscopy, spectrum analysis by Robert Bunsen and Gustav Kirchhoff (1859–1860), Crookes applied the new technique to the study of selenium compounds. Bunsen and Kirchhoff had previously used spectroscopy as a means of chemical analysis to discover caesium and rubidium. In 1861, Crookes used this process to discover thallium in some seleniferous deposits. He continued work on that new element, isolated it, studied its properties, and in 1873 determined its atomic weight. During his studies of thallium, Crookes discovered the principle of the Crookes radiometer, a device that converts light radiation into rotary motion. The principle of this radiometer has found numerous applications in the development of sensitive measuring instruments.
In 1862, Alexander Parkes exhibited Parkesine, one of the earliest synthetic polymers, at the International Exhibition in London. This discovery formed the foundation of the modern plastics industry. In 1864, Cato Maximilian Guldberg and Peter Waage, building on Claude Louis Berthollet's ideas, proposed the law of mass action. In 1865, Johann Josef Loschmidt determined the number of molecules in a mole (unit), mole, later named Avogadro constant, Avogadro's number.
In 1865, August Kekulé, based partially on the work of Loschmidt and others, established the structure of benzene as a six carbon ring with alternating single and double bonds. Kekulé's novel proposal for benzene's cyclic structure was much contested but was never replaced by a superior theory. This theory provided the scientific basis for the dramatic expansion of the German chemical industry in the last third of the 19th century. Kekulé is also famous for having clarified the nature of aromatic compounds, which are compounds based on the benzene molecule. In 1865, Adolf von Baeyer began work on indigo dye, a milestone in modern industrial organic chemistry which revolutionized the dye industry.
Swedish chemist and inventor Alfred Nobel found that when nitroglycerin was incorporated in an absorbent inert substance like ''kieselguhr'' (diatomaceous earth) it became safer and more convenient to handle, and this mixture he patented in 1867 as dynamite. Nobel later on combined nitroglycerin with various nitrocellulose compounds, similar to collodion, but settled on a more efficient recipe combining another nitrate explosive, and obtained a transparent, jelly-like substance, which was a more powerful explosive than dynamite. Gelignite, or blasting gelatin, as it was named, was patented in 1876; and was followed by a host of similar combinations, modified by the addition of potassium nitrate and various other substances.
Mendeleev's periodic table
 An important breakthrough in making sense of the list of known chemical elements (as well as in understanding the internal structure of atoms) was Dmitri Mendeleev's development of the first modern periodic table, or the periodic classification of the elements. Mendeleev, a Russian chemist, felt that there was some type of order to the elements and he spent more than thirteen years of his life collecting data and assembling the concept, initially with the idea of resolving some of the disorder in the field for his students. Mendeleev found that, when all the known chemical elements were arranged in order of increasing atomic weight, the resulting table displayed a recurring pattern, or periodicity, of properties within groups of elements. Mendeleev's law allowed him to build up a systematic periodic table of all the 66 elements then known based on atomic mass, which he published in ''Principles of Chemistry'' in 1869. His first Periodic Table was compiled on the basis of arranging the elements in ascending order of atomic weight and grouping them by similarity of properties.
Mendeleev had such faith in the validity of the periodic law that he proposed changes to the generally accepted values for the atomic weight of a few elements and, in his version of the periodic table of 1871, predicted the locations within the table of unknown elements together with their properties. He even predicted the likely properties of three yet-to-be-discovered elements, which he called Mendeleev's predicted elements, ekaboron (Eb), ekaaluminium (Ea), and ekasilicon (Es), which proved to be good predictors of the properties of scandium, gallium, and germanium, respectively, which each fill the spot in the periodic table assigned by Mendeleev.
At first the periodic system did not raise interest among chemists. However, with the discovery of the predicted elements, notably gallium in 1875, scandium in 1879, and germanium in 1886, it began to win wide acceptance. The subsequent proof of many of his predictions within his lifetime brought fame to Mendeleev as the founder of the periodic law. This organization surpassed earlier attempts at classification by Alexandre-Émile Béguyer de Chancourtois, who published the telluric helix, an early, three-dimensional version of the periodic table of the elements in 1862, John Alexander Reina Newlands, John Newlands, who proposed the law of octaves (a precursor to the periodic law) in 1864, and Julius Lothar Meyer, Lothar Meyer, who developed an early version of the periodic table with 28 elements organized by valence (chemistry), valence in 1864. Mendeleev's table did not include any of the noble gases, however, which had not yet been discovered. Gradually the periodic law and table became the framework for a great part of chemical theory. By the time Mendeleev died in 1907, he enjoyed international recognition and had received distinctions and awards from many countries.
In 1873, Jacobus Henricus van 't Hoff and Joseph Achille Le Bel, working independently, developed a model of chemical bonds, chemical bonding that explained the chirality experiments of Pasteur and provided a physical cause for optical activity in chiral compounds. van 't Hoff's publication, called V''oorstel tot Uitbreiding der Tegenwoordige in de Scheikunde gebruikte Structuurformules in de Ruimte'', etc. (Proposal for the development of 3-dimensional chemical structural formulae) and consisting of twelve pages of text and one page of diagrams, gave the impetus to the development of stereochemistry. The concept of the "asymmetrical carbon atom", dealt with in this publication, supplied an explanation of the occurrence of numerous isomers, inexplicable by means of the then current structural formulae. At the same time he pointed out the existence of relationship between optical activity and the presence of an asymmetrical carbon atom.
An important breakthrough in making sense of the list of known chemical elements (as well as in understanding the internal structure of atoms) was Dmitri Mendeleev's development of the first modern periodic table, or the periodic classification of the elements. Mendeleev, a Russian chemist, felt that there was some type of order to the elements and he spent more than thirteen years of his life collecting data and assembling the concept, initially with the idea of resolving some of the disorder in the field for his students. Mendeleev found that, when all the known chemical elements were arranged in order of increasing atomic weight, the resulting table displayed a recurring pattern, or periodicity, of properties within groups of elements. Mendeleev's law allowed him to build up a systematic periodic table of all the 66 elements then known based on atomic mass, which he published in ''Principles of Chemistry'' in 1869. His first Periodic Table was compiled on the basis of arranging the elements in ascending order of atomic weight and grouping them by similarity of properties.
Mendeleev had such faith in the validity of the periodic law that he proposed changes to the generally accepted values for the atomic weight of a few elements and, in his version of the periodic table of 1871, predicted the locations within the table of unknown elements together with their properties. He even predicted the likely properties of three yet-to-be-discovered elements, which he called Mendeleev's predicted elements, ekaboron (Eb), ekaaluminium (Ea), and ekasilicon (Es), which proved to be good predictors of the properties of scandium, gallium, and germanium, respectively, which each fill the spot in the periodic table assigned by Mendeleev.
At first the periodic system did not raise interest among chemists. However, with the discovery of the predicted elements, notably gallium in 1875, scandium in 1879, and germanium in 1886, it began to win wide acceptance. The subsequent proof of many of his predictions within his lifetime brought fame to Mendeleev as the founder of the periodic law. This organization surpassed earlier attempts at classification by Alexandre-Émile Béguyer de Chancourtois, who published the telluric helix, an early, three-dimensional version of the periodic table of the elements in 1862, John Alexander Reina Newlands, John Newlands, who proposed the law of octaves (a precursor to the periodic law) in 1864, and Julius Lothar Meyer, Lothar Meyer, who developed an early version of the periodic table with 28 elements organized by valence (chemistry), valence in 1864. Mendeleev's table did not include any of the noble gases, however, which had not yet been discovered. Gradually the periodic law and table became the framework for a great part of chemical theory. By the time Mendeleev died in 1907, he enjoyed international recognition and had received distinctions and awards from many countries.
In 1873, Jacobus Henricus van 't Hoff and Joseph Achille Le Bel, working independently, developed a model of chemical bonds, chemical bonding that explained the chirality experiments of Pasteur and provided a physical cause for optical activity in chiral compounds. van 't Hoff's publication, called V''oorstel tot Uitbreiding der Tegenwoordige in de Scheikunde gebruikte Structuurformules in de Ruimte'', etc. (Proposal for the development of 3-dimensional chemical structural formulae) and consisting of twelve pages of text and one page of diagrams, gave the impetus to the development of stereochemistry. The concept of the "asymmetrical carbon atom", dealt with in this publication, supplied an explanation of the occurrence of numerous isomers, inexplicable by means of the then current structural formulae. At the same time he pointed out the existence of relationship between optical activity and the presence of an asymmetrical carbon atom.
Josiah Willard Gibbs
 American mathematical physicist Josiah Willard Gibbs, J. Willard Gibbs's work on the applications of thermodynamics was instrumental in transforming physical chemistry into a rigorous deductive science. During the years from 1876 to 1878, Gibbs worked on the principles of thermodynamics, applying them to the complex processes involved in chemical reactions. He discovered the concept of chemical potential, or the "fuel" that makes chemical reactions work. In 1876 he published his most famous contribution, "On the Equilibrium of Heterogeneous Substances", a compilation of his work on thermodynamics and physical chemistry which laid out the concept of Thermodynamic free energy, free energy to explain the physical basis of chemical equilibria. In these essays were the beginnings of Gibbs’ theories of phases of matter: he considered each state of matter a phase, and each substance a component. Gibbs took all of the variables involved in a chemical reaction – temperature, pressure, energy, volume, and entropy – and included them in one simple equation known as Gibbs' phase rule.
Within this paper was perhaps his most outstanding contribution, the introduction of the concept of free energy, now universally called Gibbs free energy in his honor. The Gibbs free energy relates the tendency of a physical or chemical system to simultaneously lower its energy and increase its disorder, or entropy, in a spontaneous natural process. Gibbs's approach allows a researcher to calculate the change in free energy in the process, such as in a chemical reaction, and how fast it will happen. Since virtually all chemical processes and many physical ones involve such changes, his work has significantly impacted both the theoretical and experiential aspects of these sciences. In 1877, Ludwig Boltzmann established statistical derivations of many important physical and chemical concepts, including entropy, and distributions of molecular velocities in the gas phase. Together with Boltzmann and James Clerk Maxwell, Gibbs created a new branch of theoretical physics called statistical mechanics (a term that he coined), explaining the laws of thermodynamics as consequences of the statistical properties of large ensembles of particles. Gibbs also worked on the application of Maxwell's equations to problems in physical optics. Gibbs's derivation of the phenomenological laws of thermodynamics from the statistical properties of systems with many particles was presented in his highly influential textbook ''Elementary Principles in Statistical Mechanics'', published in 1902, a year before his death. In that work, Gibbs reviewed the relationship between the laws of thermodynamics and the statistical theory of molecular motions. The overshooting of the original function by partial sums of Fourier series at points of discontinuity is known as the Gibbs phenomenon.
American mathematical physicist Josiah Willard Gibbs, J. Willard Gibbs's work on the applications of thermodynamics was instrumental in transforming physical chemistry into a rigorous deductive science. During the years from 1876 to 1878, Gibbs worked on the principles of thermodynamics, applying them to the complex processes involved in chemical reactions. He discovered the concept of chemical potential, or the "fuel" that makes chemical reactions work. In 1876 he published his most famous contribution, "On the Equilibrium of Heterogeneous Substances", a compilation of his work on thermodynamics and physical chemistry which laid out the concept of Thermodynamic free energy, free energy to explain the physical basis of chemical equilibria. In these essays were the beginnings of Gibbs’ theories of phases of matter: he considered each state of matter a phase, and each substance a component. Gibbs took all of the variables involved in a chemical reaction – temperature, pressure, energy, volume, and entropy – and included them in one simple equation known as Gibbs' phase rule.
Within this paper was perhaps his most outstanding contribution, the introduction of the concept of free energy, now universally called Gibbs free energy in his honor. The Gibbs free energy relates the tendency of a physical or chemical system to simultaneously lower its energy and increase its disorder, or entropy, in a spontaneous natural process. Gibbs's approach allows a researcher to calculate the change in free energy in the process, such as in a chemical reaction, and how fast it will happen. Since virtually all chemical processes and many physical ones involve such changes, his work has significantly impacted both the theoretical and experiential aspects of these sciences. In 1877, Ludwig Boltzmann established statistical derivations of many important physical and chemical concepts, including entropy, and distributions of molecular velocities in the gas phase. Together with Boltzmann and James Clerk Maxwell, Gibbs created a new branch of theoretical physics called statistical mechanics (a term that he coined), explaining the laws of thermodynamics as consequences of the statistical properties of large ensembles of particles. Gibbs also worked on the application of Maxwell's equations to problems in physical optics. Gibbs's derivation of the phenomenological laws of thermodynamics from the statistical properties of systems with many particles was presented in his highly influential textbook ''Elementary Principles in Statistical Mechanics'', published in 1902, a year before his death. In that work, Gibbs reviewed the relationship between the laws of thermodynamics and the statistical theory of molecular motions. The overshooting of the original function by partial sums of Fourier series at points of discontinuity is known as the Gibbs phenomenon.
Late 19th century
German engineer Carl von Linde's invention of a continuous process of liquefying gases in large quantities formed a basis for the modern technology of refrigeration and provided both impetus and means for conducting scientific research at low temperatures and very high vacuums. He developed a dimethyl ether refrigerator (1874) and an ammonia refrigerator (1876). Though other refrigeration units had been developed earlier, Linde's were the first to be designed with the aim of precise calculations of efficiency. In 1895 he set up a large-scale plant for the production of liquid air. Six years later he developed a method for separating pure liquid oxygen from liquid air that resulted in widespread industrial conversion to processes utilizing oxygen (e.g., in steel
Steel is an alloy made up of iron with added carbon to improve its strength and fracture resistance compared to other forms of iron. Many other elements may be present or added. Stainless steels that are corrosion- and oxidation-resistant ty ...
manufacture).
In 1883, Svante Arrhenius developed an ion theory to explain conductivity in electrolyte
An electrolyte is a medium containing ions that is electrically conducting through the movement of those ions, but not conducting electrons. This includes most soluble salts, acids, and bases dissolved in a polar solvent, such as water. Upon dis ...
s. In 1884, Jacobus Henricus van 't Hoff published ''Études de Dynamique chimique'' (Studies in Dynamic Chemistry), a seminal study on chemical kinetics. In this work, van 't Hoff entered for the first time the field of physical chemistry. Of great importance was his development of the general thermodynamic relationship between the heat of conversion and the displacement of the equilibrium as a result of temperature variation. At constant volume, the equilibrium in a system will tend to shift in such a direction as to oppose the temperature change which is imposed upon the system. Thus, lowering the temperature results in heat development while increasing the temperature results in heat absorption. This principle of mobile equilibrium was subsequently (1885) put in a general form by Henry Louis Le Chatelier, who extended the principle to include compensation, by change of volume, for imposed pressure changes. The van 't Hoff-Le Chatelier principle, or simply Le Chatelier's principle, explains the response of dynamic equilibrium, dynamic chemical equilibrium, chemical equilibria to external stresses.
In 1884, Hermann Emil Fischer proposed the structure of purine, a key structure in many biomolecules, which he later synthesized in 1898. He also began work on the chemistry of glucose and related sugars. In 1885, Eugen Goldstein named the cathode ray, later discovered to be composed of electrons, and the canal ray, later discovered to be positive hydrogen ions that had been stripped of their electrons in a cathode ray tube; these would later be named protons. The year 1885 also saw the publishing of J. H. van 't Hoff's ''L'Équilibre chimique dans les Systèmes gazeux ou dissous à I'État dilué'' (Chemical equilibria in gaseous systems or strongly diluted solutions), which dealt with this theory of dilute solutions. Here he demonstrated that the "osmotic pressure" in solutions which are sufficiently dilute is proportionate to the concentration and the absolute temperature so that this pressure can be represented by a formula that only deviates from the formula for gas pressure by a coefficient i. He also determined the value of ''i'' by various methods, for example by means of the vapor pressure and François-Marie Raoult's results on the lowering of the freezing point. Thus van 't Hoff was able to prove that thermodynamic laws are not only valid for gases, but also for dilute solutions. His pressure laws, given general validity by the electrolytic dissociation theory of Arrhenius (1884–1887) – the first foreigner who came to work with him in Amsterdam (1888) – are considered the most comprehensive and important in the realm of natural sciences. In 1893, Alfred Werner discovered the octahedral structure of cobalt complexes, thus establishing the field of coordination chemistry.
Ramsay's discovery of the noble gases
The most celebrated discoveries of Scottish chemist William Ramsay were made in inorganic chemistry. Ramsay was intrigued by the British physicist John Strutt, 3rd Baron Rayleigh's 1892 discovery that the atomic weight of nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
found in chemical compounds was lower than that of nitrogen found in the atmosphere. He ascribed this discrepancy to a light gas included in chemical compounds of nitrogen, while Ramsay suspected a hitherto undiscovered heavy gas in atmospheric nitrogen. Using two different methods to remove all known gases from air, Ramsay and Lord Rayleigh were able to announce in 1894 that they had found a monatomic, chemically inert gaseous element that constituted nearly 1 percent of the atmosphere; they named it argon.
 The following year, Ramsay liberated another inert gas from a mineral called cleveite; this proved to be helium, previously known only in the solar spectrum. In his book ''The Gases of the Atmosphere'' (1896), Ramsay showed that the positions of helium and argon in the periodic table of elements indicated that at least three more noble gases might exist. In 1898 Ramsay and the British chemist Morris Travers, Morris W. Travers isolated these elements—called neon, krypton, and xenon—from air and brought them to a liquid state at low temperature and high pressure. Sir William Ramsay worked with Frederick Soddy to demonstrate, in 1903, that alpha particles (helium nuclei) were continually produced during the radioactive decay of a sample of radium. Ramsay was awarded the 1904 Nobel Prize for Chemistry in recognition of "services in the discovery of the inert gaseous elements in the air, and his determination of their place in the periodic system."
In 1897, J. J. Thomson discovered the electron using the cathode ray tube. In 1898, Wilhelm Wien demonstrated that canal rays (streams of positive ions) can be deflected by magnetic fields and that the amount of deflection is proportional to the mass-to-charge ratio. This discovery would lead to the analytical chemistry, analytical technique known as mass spectrometry in 1912.
The following year, Ramsay liberated another inert gas from a mineral called cleveite; this proved to be helium, previously known only in the solar spectrum. In his book ''The Gases of the Atmosphere'' (1896), Ramsay showed that the positions of helium and argon in the periodic table of elements indicated that at least three more noble gases might exist. In 1898 Ramsay and the British chemist Morris Travers, Morris W. Travers isolated these elements—called neon, krypton, and xenon—from air and brought them to a liquid state at low temperature and high pressure. Sir William Ramsay worked with Frederick Soddy to demonstrate, in 1903, that alpha particles (helium nuclei) were continually produced during the radioactive decay of a sample of radium. Ramsay was awarded the 1904 Nobel Prize for Chemistry in recognition of "services in the discovery of the inert gaseous elements in the air, and his determination of their place in the periodic system."
In 1897, J. J. Thomson discovered the electron using the cathode ray tube. In 1898, Wilhelm Wien demonstrated that canal rays (streams of positive ions) can be deflected by magnetic fields and that the amount of deflection is proportional to the mass-to-charge ratio. This discovery would lead to the analytical chemistry, analytical technique known as mass spectrometry in 1912.
Marie and Pierre Curie
 Marie Curie, Marie Skłodowska-Curie was a Polish-born French physicist and chemist who is famous for her pioneering research on radioactive decay, radioactivity. She and her husband are considered to have laid the cornerstone of the nuclear age with their research on radioactivity. Marie was fascinated with the work of Henri Becquerel, a French physicist who discovered in 1896 that uranium casts off rays similar to the X-rays discovered by Wilhelm Röntgen. Marie Curie began studying uranium in late 1897 and theorized, according to a 1904 article she wrote for Century magazine, "that the emission of rays by the compounds of uranium is a property of the metal itself—that it is an atomic property of the element uranium independent of its chemical or physical state." Curie took Becquerel's work a few steps further, conducting her own experiments on uranium rays. She discovered that the rays remained constant, no matter the condition or form of the uranium. The rays, she theorized, came from the element's atomic structure. This revolutionary idea created the field of atomic physics and the Curies coined the word ''radioactivity'' to describe the phenomena.
Marie Curie, Marie Skłodowska-Curie was a Polish-born French physicist and chemist who is famous for her pioneering research on radioactive decay, radioactivity. She and her husband are considered to have laid the cornerstone of the nuclear age with their research on radioactivity. Marie was fascinated with the work of Henri Becquerel, a French physicist who discovered in 1896 that uranium casts off rays similar to the X-rays discovered by Wilhelm Röntgen. Marie Curie began studying uranium in late 1897 and theorized, according to a 1904 article she wrote for Century magazine, "that the emission of rays by the compounds of uranium is a property of the metal itself—that it is an atomic property of the element uranium independent of its chemical or physical state." Curie took Becquerel's work a few steps further, conducting her own experiments on uranium rays. She discovered that the rays remained constant, no matter the condition or form of the uranium. The rays, she theorized, came from the element's atomic structure. This revolutionary idea created the field of atomic physics and the Curies coined the word ''radioactivity'' to describe the phenomena.
 Pierre and Marie further explored radioactivity by working to separate the substances in uranium ores and then using the electrometer to make radiation measurements to ‘trace’ the minute amount of unknown radioactive element among the fractions that resulted. Working with the mineral Uraninite, pitchblende, the pair discovered a new radioactive element in 1898. They named the element polonium, after Marie's native country of Poland. On December 21, 1898, the Curies detected the presence of another radioactive material in the pitchblende. They presented this finding to the French Academy of Sciences on December 26, proposing that the new element be called radium. The Curies then went to work isolating polonium and radium from naturally occurring compounds to prove that they were new elements. In 1902, the Curies announced that they had produced a decigram of pure radium, demonstrating its existence as a unique chemical element. While it took three years for them to isolate radium, they were never able to isolate polonium. Along with the discovery of two new elements and finding techniques for isolating radioactive isotopes, Curie oversaw the world's first studies into the treatment of neoplasms, using radioactive isotopes. With Henri Becquerel and her husband, Pierre Curie, she was awarded the 1903 Nobel Prize for Physics. She was the sole winner of the 1911 Nobel Prize for Chemistry. She was the first woman to win a Nobel Prize, and she is the only woman to win the award in two different fields.
While working with Marie to extract pure substances from ores, an undertaking that really required industrial resources but that they achieved in relatively primitive conditions, Pierre himself concentrated on the physical study (including luminous and chemical effects) of the new radiations. Through the action of magnetic fields on the rays given out by the radium, he proved the existence of particles that were electrically positive, negative, and neutral; these Ernest Rutherford was afterward to call alpha, beta, and gamma rays. Pierre then studied these radiations by calorimetry and also observed the physiological effects of radium, thus opening the way to radium therapy. Among Pierre Curie's discoveries were that ferromagnetic substances exhibited a critical temperature transition, above which the substances lost their ferromagnetic behavior – this is known as the "Curie temperature, Curie point." He was elected to the Academy of Sciences (1905), having in 1903 jointly with Marie received the Royal Society's prestigious Davy Medal and jointly with her and Becquerel the Nobel Prize for Physics. He was run over by a carriage in the rue Dauphine in Paris in 1906 and died instantly. His complete works were published in 1908.
Pierre and Marie further explored radioactivity by working to separate the substances in uranium ores and then using the electrometer to make radiation measurements to ‘trace’ the minute amount of unknown radioactive element among the fractions that resulted. Working with the mineral Uraninite, pitchblende, the pair discovered a new radioactive element in 1898. They named the element polonium, after Marie's native country of Poland. On December 21, 1898, the Curies detected the presence of another radioactive material in the pitchblende. They presented this finding to the French Academy of Sciences on December 26, proposing that the new element be called radium. The Curies then went to work isolating polonium and radium from naturally occurring compounds to prove that they were new elements. In 1902, the Curies announced that they had produced a decigram of pure radium, demonstrating its existence as a unique chemical element. While it took three years for them to isolate radium, they were never able to isolate polonium. Along with the discovery of two new elements and finding techniques for isolating radioactive isotopes, Curie oversaw the world's first studies into the treatment of neoplasms, using radioactive isotopes. With Henri Becquerel and her husband, Pierre Curie, she was awarded the 1903 Nobel Prize for Physics. She was the sole winner of the 1911 Nobel Prize for Chemistry. She was the first woman to win a Nobel Prize, and she is the only woman to win the award in two different fields.
While working with Marie to extract pure substances from ores, an undertaking that really required industrial resources but that they achieved in relatively primitive conditions, Pierre himself concentrated on the physical study (including luminous and chemical effects) of the new radiations. Through the action of magnetic fields on the rays given out by the radium, he proved the existence of particles that were electrically positive, negative, and neutral; these Ernest Rutherford was afterward to call alpha, beta, and gamma rays. Pierre then studied these radiations by calorimetry and also observed the physiological effects of radium, thus opening the way to radium therapy. Among Pierre Curie's discoveries were that ferromagnetic substances exhibited a critical temperature transition, above which the substances lost their ferromagnetic behavior – this is known as the "Curie temperature, Curie point." He was elected to the Academy of Sciences (1905), having in 1903 jointly with Marie received the Royal Society's prestigious Davy Medal and jointly with her and Becquerel the Nobel Prize for Physics. He was run over by a carriage in the rue Dauphine in Paris in 1906 and died instantly. His complete works were published in 1908.
Ernest Rutherford
 New Zealand-born chemist and physicist Ernest Rutherford is considered to be "the father of nuclear physics." Rutherford is best known for devising the names alpha particle, alpha, beta particle, beta, and gamma ray, gamma to classify various forms of radioactive "rays" which were poorly understood at his time (alpha and beta rays are particle beams, while gamma rays are a form of high-energy electromagnetic radiation). Rutherford deflected alpha rays with both electric and magnetic fields in 1903. Working with Frederick Soddy, Rutherford explained that radioactivity is due to the nuclear transmutation, transmutation of elements, now known to involve nuclear reactions.
New Zealand-born chemist and physicist Ernest Rutherford is considered to be "the father of nuclear physics." Rutherford is best known for devising the names alpha particle, alpha, beta particle, beta, and gamma ray, gamma to classify various forms of radioactive "rays" which were poorly understood at his time (alpha and beta rays are particle beams, while gamma rays are a form of high-energy electromagnetic radiation). Rutherford deflected alpha rays with both electric and magnetic fields in 1903. Working with Frederick Soddy, Rutherford explained that radioactivity is due to the nuclear transmutation, transmutation of elements, now known to involve nuclear reactions.
 He also observed that the intensity of radioactivity of a radioactive element decreases over a unique and regular amount of time until a point of stability, and he named the halving time the "half-life." In 1901 and 1902 he worked with Frederick Soddy to prove that atoms of one radioactive element would spontaneously turn into another, by expelling a piece of the atom at high velocity. In 1906 at the University of Manchester, Rutherford oversaw an experiment conducted by his students Hans Geiger (known for the Geiger counter) and Ernest Marsden. In the Geiger–Marsden experiment, a beam of alpha particles, generated by the radioactive decay of radon, was directed normally onto a sheet of very thin gold foil in an evacuated chamber. Under the prevailing plum pudding model, the alpha particles should all have passed through the foil and hit the detector screen, or have been deflected by, at most, a few degrees.
However, the actual results surprised Rutherford. Although many of the alpha particles did pass through as expected, many others were deflected at small angles while others were reflected back to the alpha source. They observed that a very small percentage of particles were deflected through angles much larger than 90 degrees. The gold foil experiment showed large deflections for a small fraction of incident particles. Rutherford realized that, because some of the alpha particles were deflected or reflected, the atom had a concentrated centre of positive charge and of relatively large mass – Rutherford later termed this positive center the "atomic nucleus". The alpha particles had either hit the positive centre directly or passed by it close enough to be affected by its positive charge. Since many other particles passed through the gold foil, the positive centre would have to be a relatively small size compared to the rest of the atom – meaning that the atom is mostly open space. From his results, Rutherford developed a model of the atom that was similar to the solar system, known as the Rutherford model. Like planets, electrons orbited a central, sun-like nucleus. For his work with radiation and the atomic nucleus, Rutherford received the 1908 Nobel Prize in Chemistry.
He also observed that the intensity of radioactivity of a radioactive element decreases over a unique and regular amount of time until a point of stability, and he named the halving time the "half-life." In 1901 and 1902 he worked with Frederick Soddy to prove that atoms of one radioactive element would spontaneously turn into another, by expelling a piece of the atom at high velocity. In 1906 at the University of Manchester, Rutherford oversaw an experiment conducted by his students Hans Geiger (known for the Geiger counter) and Ernest Marsden. In the Geiger–Marsden experiment, a beam of alpha particles, generated by the radioactive decay of radon, was directed normally onto a sheet of very thin gold foil in an evacuated chamber. Under the prevailing plum pudding model, the alpha particles should all have passed through the foil and hit the detector screen, or have been deflected by, at most, a few degrees.
However, the actual results surprised Rutherford. Although many of the alpha particles did pass through as expected, many others were deflected at small angles while others were reflected back to the alpha source. They observed that a very small percentage of particles were deflected through angles much larger than 90 degrees. The gold foil experiment showed large deflections for a small fraction of incident particles. Rutherford realized that, because some of the alpha particles were deflected or reflected, the atom had a concentrated centre of positive charge and of relatively large mass – Rutherford later termed this positive center the "atomic nucleus". The alpha particles had either hit the positive centre directly or passed by it close enough to be affected by its positive charge. Since many other particles passed through the gold foil, the positive centre would have to be a relatively small size compared to the rest of the atom – meaning that the atom is mostly open space. From his results, Rutherford developed a model of the atom that was similar to the solar system, known as the Rutherford model. Like planets, electrons orbited a central, sun-like nucleus. For his work with radiation and the atomic nucleus, Rutherford received the 1908 Nobel Prize in Chemistry.
20th century
 In 1903, Mikhail Tsvet invented chromatography, an important analytic technique. In 1904, Hantaro Nagaoka proposed an early nuclear model of the atom, where electrons orbit a dense massive nucleus. In 1905, Fritz Haber and Carl Bosch developed the Haber process for making
In 1903, Mikhail Tsvet invented chromatography, an important analytic technique. In 1904, Hantaro Nagaoka proposed an early nuclear model of the atom, where electrons orbit a dense massive nucleus. In 1905, Fritz Haber and Carl Bosch developed the Haber process for making ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
, a milestone in industrial chemistry with deep consequences in agriculture. The Haber process, or Haber-Bosch process, combined nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
and hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
to form ammonia in industrial quantities for the production of fertilizer and munitions. The food production for half the world's current population depends on this method for producing fertilizer. Haber, along with Max Born, proposed the Born–Haber cycle as a method for evaluating the lattice energy of an ionic solid. Haber has also been described as the "father of chemical warfare" for his work developing and deploying chlorine and other poisonous gases during World War I.
 In 1905, Albert Einstein explained Brownian motion in a way that definitively proved atomic theory. Leo Baekeland invented bakelite, one of the first commercially successful plastics. In 1909, American physicist Robert Andrews Millikan – who had studied in Europe under Walther Nernst and Max Planck – measured the charge of individual electrons with unprecedented accuracy through the oil drop experiment, in which he measured the electric charges on tiny falling water (and later oil) droplets. His study established that any particular droplet's electrical charge is a multiple of a definite, fundamental value — the electron's charge — and thus a confirmation that all electrons have the same charge and mass. Beginning in 1912, he spent several years investigating and finally proving Albert Einstein's proposed linear relationship between energy and frequency, and providing the first direct Photoelectric effect, photoelectric support for Planck's constant. In 1923 Millikan was awarded the Nobel Prize for Physics.
In 1909, S. P. L. Sørensen invented the pH concept and developed methods for measuring acidity. In 1911, Antonius Van den Broek proposed the idea that the elements on the periodic table are more properly organized by positive nuclear charge rather than atomic weight. In 1911, the first Solvay Conference was held in Brussels, bringing together most of the most prominent scientists of the day. In 1912, William Henry Bragg and William Lawrence Bragg proposed Bragg's law and established the field of X-ray crystallography, an important tool for elucidating the crystal structure of substances. In 1912, Peter Debye used the concept of a molecular dipole to describe asymmetric charge distribution in some molecules.
In 1905, Albert Einstein explained Brownian motion in a way that definitively proved atomic theory. Leo Baekeland invented bakelite, one of the first commercially successful plastics. In 1909, American physicist Robert Andrews Millikan – who had studied in Europe under Walther Nernst and Max Planck – measured the charge of individual electrons with unprecedented accuracy through the oil drop experiment, in which he measured the electric charges on tiny falling water (and later oil) droplets. His study established that any particular droplet's electrical charge is a multiple of a definite, fundamental value — the electron's charge — and thus a confirmation that all electrons have the same charge and mass. Beginning in 1912, he spent several years investigating and finally proving Albert Einstein's proposed linear relationship between energy and frequency, and providing the first direct Photoelectric effect, photoelectric support for Planck's constant. In 1923 Millikan was awarded the Nobel Prize for Physics.
In 1909, S. P. L. Sørensen invented the pH concept and developed methods for measuring acidity. In 1911, Antonius Van den Broek proposed the idea that the elements on the periodic table are more properly organized by positive nuclear charge rather than atomic weight. In 1911, the first Solvay Conference was held in Brussels, bringing together most of the most prominent scientists of the day. In 1912, William Henry Bragg and William Lawrence Bragg proposed Bragg's law and established the field of X-ray crystallography, an important tool for elucidating the crystal structure of substances. In 1912, Peter Debye used the concept of a molecular dipole to describe asymmetric charge distribution in some molecules.
Niels Bohr
 In 1913, Niels Bohr, a Danish physicist, introduced the concepts of quantum mechanics to atomic structure by proposing what is now known as the Bohr model of the atom, where electrons exist only in strictly defined circular orbits around the nucleus similar to rungs on a ladder. The Bohr Model is a planetary model in which the negatively charged electrons orbit a small, positively charged nucleus similar to the planets orbiting the Sun (except that the orbits are not planar) – the gravitational force of the solar system is mathematically akin to the attractive Coulomb (electrical) force between the positively charged nucleus and the negatively charged electrons.
In the Bohr model, however, electrons orbit the nucleus in orbits that have a set size and energy – the energy levels are said to be ''quantized'', which means that only certain orbits with certain radii are allowed; orbits in between simply don't exist. The energy of the orbit is related to its size – that is, the lowest energy is found in the smallest orbit. Bohr also postulated that electromagnetic radiation is absorbed or emitted when an electron moves from one orbit to another. Because only certain electron orbits are permitted, the emission of light accompanying a jump of an electron from an excited energy state to ground state produces a unique emission spectrum for each element. Bohr later received the Nobel Prize in physics for this work.
Niels Bohr also worked on the principle of Complementarity (physics), complementarity, which states that an electron can be interpreted in two mutually exclusive and valid ways. Electrons can be interpreted as wave or particle models. His hypothesis was that an incoming particle would strike the nucleus and create an excited compound nucleus. This formed the basis of his Semi-empirical mass formula, liquid drop model and later provided a theory base for nuclear fission after its discovery of nuclear fission, discovery by chemists Otto Hahn and Fritz Strassman, and explanation and naming by physicists Lise Meitner and Otto Frisch.
In 1913, Niels Bohr, a Danish physicist, introduced the concepts of quantum mechanics to atomic structure by proposing what is now known as the Bohr model of the atom, where electrons exist only in strictly defined circular orbits around the nucleus similar to rungs on a ladder. The Bohr Model is a planetary model in which the negatively charged electrons orbit a small, positively charged nucleus similar to the planets orbiting the Sun (except that the orbits are not planar) – the gravitational force of the solar system is mathematically akin to the attractive Coulomb (electrical) force between the positively charged nucleus and the negatively charged electrons.
In the Bohr model, however, electrons orbit the nucleus in orbits that have a set size and energy – the energy levels are said to be ''quantized'', which means that only certain orbits with certain radii are allowed; orbits in between simply don't exist. The energy of the orbit is related to its size – that is, the lowest energy is found in the smallest orbit. Bohr also postulated that electromagnetic radiation is absorbed or emitted when an electron moves from one orbit to another. Because only certain electron orbits are permitted, the emission of light accompanying a jump of an electron from an excited energy state to ground state produces a unique emission spectrum for each element. Bohr later received the Nobel Prize in physics for this work.
Niels Bohr also worked on the principle of Complementarity (physics), complementarity, which states that an electron can be interpreted in two mutually exclusive and valid ways. Electrons can be interpreted as wave or particle models. His hypothesis was that an incoming particle would strike the nucleus and create an excited compound nucleus. This formed the basis of his Semi-empirical mass formula, liquid drop model and later provided a theory base for nuclear fission after its discovery of nuclear fission, discovery by chemists Otto Hahn and Fritz Strassman, and explanation and naming by physicists Lise Meitner and Otto Frisch.
 In 1913, Henry Moseley, working from Van den Broek's earlier idea, introduced the concept of atomic number to fix some inadequacies of Mendeleev's periodic table, which had been based on atomic weight. The peak of Frederick Soddy's career in radiochemistry was in 1913 with his formulation of the concept of isotopes, which stated that certain elements exist in two or more forms which have different atomic weights but which are indistinguishable chemically. He is remembered for proving the existence of isotopes of certain radioactive elements, and is also credited, along with others, with the discovery of the element protactinium in 1917. In 1913, J. J. Thomson expanded on the work of Wien by showing that charged subatomic particles can be separated by their mass-to-charge ratio, a technique known as mass spectrometry.
In 1913, Henry Moseley, working from Van den Broek's earlier idea, introduced the concept of atomic number to fix some inadequacies of Mendeleev's periodic table, which had been based on atomic weight. The peak of Frederick Soddy's career in radiochemistry was in 1913 with his formulation of the concept of isotopes, which stated that certain elements exist in two or more forms which have different atomic weights but which are indistinguishable chemically. He is remembered for proving the existence of isotopes of certain radioactive elements, and is also credited, along with others, with the discovery of the element protactinium in 1917. In 1913, J. J. Thomson expanded on the work of Wien by showing that charged subatomic particles can be separated by their mass-to-charge ratio, a technique known as mass spectrometry.
Gilbert N. Lewis
American physical chemist Gilbert N. Lewis laid the foundation of valence bond theory; he was instrumental in developing a bonding theory based on the number of electrons in the outermost "valence" shell of the atom. In 1902, while Lewis was trying to explain valence to his students, he depicted atoms as constructed of a concentric series of cubes with electrons at each corner. This "cubic atom" explained the eight groups in the periodic table and represented his idea that chemical bonds are formed by electron transference to give each atom a complete set of eight outer electrons (an "octet").
Lewis's theory of chemical bonding continued to evolve and, in 1916, he published his seminal article "The Atom of the Molecule", which suggested that a chemical bond is a pair of electrons shared by two atoms. Lewis's model equated the classical chemical bond with the sharing of a pair of electrons between the two bonded atoms. Lewis introduced the "electron dot diagrams" in this paper to symbolize the electronic structures of atoms and molecules. Now known as Lewis structures, they are discussed in virtually every introductory chemistry book.
Shortly after the publication of his 1916 paper, Lewis became involved with military research. He did not return to the subject of chemical bonding until 1923, when he masterfully summarized his model in a short monograph entitled Valence and the Structure of Atoms and Molecules. His renewal of interest in this subject was largely stimulated by the activities of the American chemist and General Electric researcher Irving Langmuir, who between 1919 and 1921 popularized and elaborated Lewis's model. Langmuir subsequently introduced the term ''covalent bond''. In 1921, Otto Stern and Walther Gerlach established the concept of quantum mechanical spin in subatomic particles.
For cases where no sharing was involved, Lewis in 1923 developed the electron pair theory of acid
In computer science, ACID ( atomicity, consistency, isolation, durability) is a set of properties of database transactions intended to guarantee data validity despite errors, power failures, and other mishaps. In the context of databases, a sequ ...
s and base (chemistry), base: Lewis redefined an acid as any atom or molecule with an incomplete octet that was thus capable of accepting electrons from another atom; bases were, of course, electron donors. His theory is known as the concept of Lewis acids and bases. In 1923, G. N. Lewis and Merle Randall published ''Thermodynamics and the Free Energy of Chemical Substances'', first modern treatise on chemical thermodynamics.
The 1920s saw a rapid adoption and application of Lewis's model of the electron-pair bond in the fields of organic and coordination chemistry. In organic chemistry, this was primarily due to the efforts of the British chemists Arthur Lapworth, Robert Robinson (organic chemist), Robert Robinson, Martin Lowry, Thomas Lowry, and Christopher Ingold; while in coordination chemistry, Lewis's bonding model was promoted through the efforts of the American chemist Maurice Huggins and the British chemist Nevil Sidgwick.
Quantum mechanics
In 1924, French quantum physicist Louis de Broglie published his thesis, in which he introduced a revolutionary theory of electron waves based on wave–particle duality. In his time, the wave and particle interpretations of light and matter were seen as being at odds with one another, but de Broglie suggested that these seemingly different characteristics were instead the same behavior observed from different perspectives — that particles can behave like waves, and waves (radiation) can behave like particles. Broglie's proposal offered an explanation of the restricted motion of electrons within the atom. The first publications of Broglie's idea of "matter waves" had drawn little attention from other physicists, but a copy of his doctoral thesis chanced to reach Einstein, whose response was enthusiastic. Einstein stressed the importance of Broglie's work both explicitly and by building further on it.
In 1925, Austrian-born physicist Wolfgang Pauli developed the Pauli exclusion principle, which states that no two electrons around a single nucleus in an atom can occupy the same quantum state simultaneously, as described by four quantum numbers. Pauli made major contributions to quantum mechanics and quantum field theory – he was awarded the 1945 Nobel Prize for Physics for his discovery of the Pauli exclusion principle – as well as solid-state physics, and he successfully hypothesized the existence of the neutrino. In addition to his original work, he wrote masterful syntheses of several areas of physical theory that are considered classics of scientific literature.
In 1926 at the age of 39, Austrian theoretical physicist Erwin Schrödinger produced the papers that gave the foundations of quantum wave mechanics. In those papers he described his partial differential equation that is the basic equation of quantum mechanics and bears the same relation to the mechanics of the atom as Newton's laws of motion, Newton's equations of motion bear to planetary astronomy. Adopting a proposal made by Louis de Broglie in 1924 that particles of matter have a dual nature and in some situations act like waves, Schrödinger introduced a theory describing the behaviour of such a system by a wave equation that is now known as the Schrödinger equation. The solutions to Schrödinger's equation, unlike the solutions to Newton's equations, are wave functions that can only be related to the probable occurrence of physical events. The readily visualized sequence of events of the planetary orbits of Newton is, in quantum mechanics, replaced by the more abstract notion of probability. (This aspect of the quantum theory made Schrödinger and several other physicists profoundly unhappy, and he devoted much of his later life to formulating philosophical objections to the generally accepted interpretation of the theory that he had done so much to create.)
German theoretical physicist Werner Heisenberg was one of the key creators of quantum mechanics. In 1925, Heisenberg discovered a way to formulate quantum mechanics in terms of matrices. For that discovery, he was awarded the Nobel Prize for Physics for 1932. In 1927 he published his uncertainty principle, upon which he built his philosophy and for which he is best known. Heisenberg was able to demonstrate that if you were studying an electron in an atom you could say where it was (the electron's location) or where it was going (the electron's velocity), but it was impossible to express both at the same time. He also made important contributions to the theories of the hydrodynamics of turbulent flows, the atomic nucleus, ferromagnetism, cosmic rays, and subatomic particles, and he was instrumental in planning the first West German nuclear reactor at Karlsruhe, together with a research reactor in Munich, in 1957. Considerable controversy surrounds his work on atomic research during World War II.
Quantum chemistry
Some view the birth of quantum chemistry in the discovery of the Schrödinger equation and its application to the hydrogen atom in 1926. However, the 1927 article of Walter Heitler and Fritz London is often recognised as the first milestone in the history of quantum chemistry. This is the first application of quantum mechanics to the diatomic hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
molecule, and thus to the phenomenon of the chemical bond. In the following years much progress was accomplished by Edward Teller, Robert S. Mulliken, Max Born, J. Robert Oppenheimer, Linus Pauling, Erich Hückel, Douglas Hartree and Vladimir Aleksandrovich Fock, to cite a few.
Still, skepticism remained as to the general power of quantum mechanics applied to complex chemical systems. The situation around 1930 is described by Paul Dirac:
Hence the quantum mechanical methods developed in the 1930s and 1940s are often referred to as theoretical molecular physics, molecular or atomic physics to underline the fact that they were more the application of quantum mechanics to chemistry and spectroscopy than answers to chemically relevant questions. In 1951, a milestone article in quantum chemistry is the seminal paper of Clemens C. J. Roothaan on Roothaan equations. It opened the avenue to the solution of the self-consistent field equations for small molecules like hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, an ...
or nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
. Those computations were performed with the help of tables of integrals which were computed on the most advanced computers of the time.
In the 1940s many physicists turned from molecular physics, molecular or atomic physics to nuclear physics (like J. Robert Oppenheimer or Edward Teller). Glenn T. Seaborg was an American nuclear chemist best known for his work on isolating and identifying transuranium elements (those heavier than uranium). He shared the 1951 Nobel Prize for Chemistry with Edwin McMillan, Edwin Mattison McMillan for their independent discoveries of transuranium elements. Seaborgium was named in his honour, making him the only person, along with Albert Einstein and Yuri Oganessian, for whom a chemical element was named during his lifetime.
Molecular biology and biochemistry
By the mid 20th century, in principle, the integration of physics and chemistry was extensive, with chemical properties explained as the result of the electronic structure of the atom; Linus Pauling's book on ''The Nature of the Chemical Bond'' used the principles of quantum mechanics to deduce bond angles in ever-more complicated molecules. However, though some principles deduced from quantum mechanics were able to predict qualitatively some chemical features for biologically relevant molecules, they were, till the end of the 20th century, more a collection of rules, observations, and recipes than rigorous ab initio quantitative methods.
 This heuristic approach triumphed in 1953 when James D. Watson, James Watson and Francis Crick deduced the double helical structure of DNA by constructing models constrained by and informed by the knowledge of the chemistry of the constituent parts and the X-ray diffraction patterns obtained by Rosalind Franklin. This discovery lead to an explosion of research into the biochemistry of life.
In the same year, the Miller–Urey experiment demonstrated that basic constituents of protein, simple amino acids, could themselves be built up from simpler molecules in a simulation of primordial Process (science), processes on Earth. This first attempt by chemists to study hypothetical processes in the laboratory under controlled conditions helped kickstart bountiful research, within the natural sciences, into the origins of life.
In 1983 Kary Mullis devised a method for the in-vitro amplification of DNA, known as the polymerase chain reaction (PCR), which revolutionized the chemical processes used in the laboratory to manipulate it. PCR could be used to synthesize specific pieces of DNA and made possible the DNA sequencing, sequencing of DNA of organisms, which culminated in the huge human genome project.
An important piece in the double helix puzzle was solved by one of Pauling's students Matthew Meselson and Frank Stahl, the result of their collaboration (Meselson–Stahl experiment) has been called as "the most beautiful experiment in biology".
They used a centrifugation technique that sorted molecules according to differences in weight. Because nitrogen atoms are a component of DNA, they were labelled and therefore tracked in replication in bacteria.
This heuristic approach triumphed in 1953 when James D. Watson, James Watson and Francis Crick deduced the double helical structure of DNA by constructing models constrained by and informed by the knowledge of the chemistry of the constituent parts and the X-ray diffraction patterns obtained by Rosalind Franklin. This discovery lead to an explosion of research into the biochemistry of life.
In the same year, the Miller–Urey experiment demonstrated that basic constituents of protein, simple amino acids, could themselves be built up from simpler molecules in a simulation of primordial Process (science), processes on Earth. This first attempt by chemists to study hypothetical processes in the laboratory under controlled conditions helped kickstart bountiful research, within the natural sciences, into the origins of life.
In 1983 Kary Mullis devised a method for the in-vitro amplification of DNA, known as the polymerase chain reaction (PCR), which revolutionized the chemical processes used in the laboratory to manipulate it. PCR could be used to synthesize specific pieces of DNA and made possible the DNA sequencing, sequencing of DNA of organisms, which culminated in the huge human genome project.
An important piece in the double helix puzzle was solved by one of Pauling's students Matthew Meselson and Frank Stahl, the result of their collaboration (Meselson–Stahl experiment) has been called as "the most beautiful experiment in biology".
They used a centrifugation technique that sorted molecules according to differences in weight. Because nitrogen atoms are a component of DNA, they were labelled and therefore tracked in replication in bacteria.
Late 20th century
 In 1970, John Pople developed the Gaussian (software), Gaussian program greatly easing computational chemistry calculations. In 1971, Yves Chauvin offered an explanation of the reaction mechanism of olefin metathesis reactions. In 1975, Karl Barry Sharpless and his group discovered stereoselective redox, oxidation reactions including Sharpless epoxidation, Sharpless asymmetric dihydroxylation, and Sharpless oxyamination.
In 1985, Harold Kroto, Robert Curl and Richard Smalley discovered fullerenes, a class of large carbon molecules superficially resembling the geodesic dome designed by architect R. Buckminster Fuller. In 1991, Sumio Iijima used electron microscopy to discover a type of cylindrical fullerene known as a carbon nanotube, though earlier work had been done in the field as early as 1951. This material is an important component in the field of nanotechnology. In 1994, K. C. Nicolaou with his group and Robert A. Holton and his group, achieved the first Holton Taxol total synthesis, total synthesis of Taxol. In 1995, Eric Cornell and Carl Wieman produced the first Bose–Einstein condensate, a substance that displays quantum mechanical properties on the macroscopic scale.
In 1970, John Pople developed the Gaussian (software), Gaussian program greatly easing computational chemistry calculations. In 1971, Yves Chauvin offered an explanation of the reaction mechanism of olefin metathesis reactions. In 1975, Karl Barry Sharpless and his group discovered stereoselective redox, oxidation reactions including Sharpless epoxidation, Sharpless asymmetric dihydroxylation, and Sharpless oxyamination.
In 1985, Harold Kroto, Robert Curl and Richard Smalley discovered fullerenes, a class of large carbon molecules superficially resembling the geodesic dome designed by architect R. Buckminster Fuller. In 1991, Sumio Iijima used electron microscopy to discover a type of cylindrical fullerene known as a carbon nanotube, though earlier work had been done in the field as early as 1951. This material is an important component in the field of nanotechnology. In 1994, K. C. Nicolaou with his group and Robert A. Holton and his group, achieved the first Holton Taxol total synthesis, total synthesis of Taxol. In 1995, Eric Cornell and Carl Wieman produced the first Bose–Einstein condensate, a substance that displays quantum mechanical properties on the macroscopic scale.
Mathematics and chemistry
Before the 20th century, chemistry was defined as the science of the nature of matter and its transformations. It was therefore distinct from physics which was not concerned with such dramatic transformation of matter. Moreover, in contrast to physics, chemistry remained predominantly a descriptive and empirical science until the end of the 19th century. Though they developed a consistent quantitative foundation based on notions of atomic and molecular weights, combining proportions, and thermodynamic quantities, chemists had less use of advanced mathematics.
Scope of chemistry
As understanding of the nature of matter has evolved, so too has the self-understanding of the science of chemistry by its practitioners. This continuing historical process of evaluation includes the categories, terms, aims and scope of chemistry. Additionally, the development of the social institutions and networks which support chemical enquiry are highly significant factors that enable the production, dissemination and application of chemical knowledge. (See Philosophy of chemistry)
Chemical industry
The later part of the nineteenth century saw a huge increase in the exploitation of petroleum extracted from the earth for the production of a host of chemicals and largely replaced the use of whale oil, coal tar and naval stores used previously. Large-scale production and oil refinery, refinement of petroleum provided feedstocks for liquid fuels such as gasoline and Diesel fuel, diesel, solvents, lubricants, asphalt, waxes, and for the production of many of the common materials of the modern world, such as synthetic fibers, plastics, paints, detergents, pharmaceuticals, adhesives and ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
as fertilizer and for other uses. Many of these required new catalysts and the utilization of chemical engineering for their cost-effective production.
In the mid-twentieth century, control of the electronic structure of semiconductor materials was made precise by the creation of large ingots of extremely pure single crystals of silicon and germanium. Accurate control of their chemical composition by doping with other elements made the production of the solid state transistor in 1951 and made possible the production of tiny integrated circuits for use in electronic devices, especially computers.
See also
Histories and timelines
* Atomic theory
* Cupellation
* History of chromatography
* History of electrochemistry
* History of the molecule
* History of molecular biology
* History of physics
* History of science and technology
* History of the periodic table
* History of thermodynamics
* History of energy
* History of molecular theory
* History of materials science
* List of years in science
* Nobel Prize in chemistry
* Timeline of atomic and subatomic physics
* Timeline of chemical elements discoveries
* Timeline of chemistry
* Timeline of materials technology
* Timeline of thermodynamics, statistical mechanics, and random processes
* The Chemical History of a Candle
* The Mystery of Matter (film), The Mystery of Matter: Search for the Elements (PBS film)
Notable chemists
''listed chronologically:''
* List of chemists
* Robert Boyle
Robert Boyle (; 25 January 1627 – 31 December 1691) was an Anglo-Irish natural philosopher, chemist, physicist, alchemist and inventor. Boyle is largely regarded today as the first modern chemist, and therefore one of the founders of ...
, 1627–1691
* Joseph Black
Joseph Black (16 April 1728 – 6 December 1799) was a Scottish physicist and chemist, known for his discoveries of magnesium, latent heat, specific heat, and carbon dioxide. He was Professor of Anatomy and Chemistry at the University of Glas ...
, 1728–1799
* Joseph Priestley
Joseph Priestley (; 24 March 1733 – 6 February 1804) was an English chemist, natural philosopher, separatist theologian, grammarian, multi-subject educator, and liberal political theorist. He published over 150 works, and conducted exp ...
, 1733–1804
* Carl Wilhelm Scheele
Carl Wilhelm Scheele (, ; 9 December 1742 – 21 May 1786) was a Swedish German pharmaceutical chemist.
Scheele discovered oxygen (although Joseph Priestley published his findings first), and identified molybdenum, tungsten, barium, hydrog ...
, 1742–1786
* Antoine Lavoisier, 1743–1794
* Alessandro Volta
Alessandro Giuseppe Antonio Anastasio Volta (, ; 18 February 1745 – 5 March 1827) was an Italian physicist, chemist and lay Catholic who was a pioneer of electricity and power who is credited as the inventor of the electric battery and the ...
, 1745–1827
* Jacques Charles, 1746–1823
* Claude Louis Berthollet
Claude Louis Berthollet (, 9 December 1748 – 6 November 1822) was a Savoyard-French chemist who became vice president of the French Senate in 1804. He is known for his scientific contributions to theory of chemical equilibria via the mecha ...
, 1748–1822
* Amedeo Avogadro, 1776–1856
* Joseph-Louis Gay-Lussac, 1778–1850
* Humphry Davy, 1778–1829
* Jöns Jacob Berzelius, inventor of modern chemical notation, 1779–1848
* Justus von Liebig, 1803–1873
* Louis Pasteur, 1822–1895
* Stanislao Cannizzaro, 1826–1910
* Friedrich August Kekulé von Stradonitz, 1829–1896
* Dmitri Mendeleev, 1834–1907
* Josiah Willard Gibbs, 1839–1903
* J. H. van 't Hoff, 1852–1911
* William Ramsay, 1852–1916
* Svante Arrhenius, 1859–1927
* Walther Nernst, 1864–1941
* Marie Curie, 1867–1934
* Gilbert N. Lewis, 1875–1946
* Otto Hahn, 1879–1968
* Irving Langmuir, 1881–1957
* Linus Pauling, 1901–1994
* Glenn T. Seaborg, 1912–1999
* Robert Burns Woodward, 1917–1979
* Frederick Sanger, 1918–2013
* Geoffrey Wilkinson, 1921–1996
* Rudolph A. Marcus, 1923–
* George Andrew Olah, 1926–2017
* Elias James Corey, 1928–
* Akira Suzuki (chemist), Akira Suzuki, 1930–
* Richard F. Heck, 1931–2015
* Harold Kroto, 1939–2016
* Jean-Marie Lehn, 1939–
* Peter Atkins, 1940–
* Barry Sharpless, 1941–
* Richard Smalley, 1943–2005
* Jean-Pierre Sauvage, 1944–
Notes
References
Selected classic papers from the history of chemistry
*Eric R. Scerri, The Periodic Table: Its Story and Its Significance, Oxford University Press, 2006.
Further reading
*
*
* John Servos, Servos, John W.
''Physical chemistry from Ostwald to Pauling : the making of a science in America''
Princeton, N.J. : Princeton University Press, 1990.
; Documentaries
* BBC (2010). ''Chemistry: A Volatile History''.
External links
ChemisLab
– Chemists of the Past
SHAC: Society for the History of Alchemy and Chemistry
{{DEFAULTSORT:History Of Chemistry
History of chemistry,
History of science, Chemistry
History of science by discipline, Chemistry
History of industries, Chemistry
 The history of chemistry represents a time span from
The history of chemistry represents a time span from  Philosophical attempts to rationalize why different substances have different properties (color, density, smell), exist in different states (gaseous, liquid, and solid), and react in a different manner when exposed to environments, for example to water or fire or temperature changes, led ancient philosophers to postulate the first theories on nature and chemistry. The history of such philosophical theories that relate to chemistry can probably be traced back to every single ancient civilization. The common aspect in all these theories was the attempt to identify a small number of primary
Philosophical attempts to rationalize why different substances have different properties (color, density, smell), exist in different states (gaseous, liquid, and solid), and react in a different manner when exposed to environments, for example to water or fire or temperature changes, led ancient philosophers to postulate the first theories on nature and chemistry. The history of such philosophical theories that relate to chemistry can probably be traced back to every single ancient civilization. The common aspect in all these theories was the attempt to identify a small number of primary  The elemental system used in medieval
The elemental system used in medieval  Alchemy is defined by the
Alchemy is defined by the  Practical attempts to improve the refining of ores and their extraction to smelt metals was an important source of information for early chemists in the 16th century, among them
Practical attempts to improve the refining of ores and their extraction to smelt metals was an important source of information for early chemists in the 16th century, among them 
 Anglo-Irish chemist
Anglo-Irish chemist  In 1702, German chemist
In 1702, German chemist  Antoine-Laurent de Lavoisier demonstrated with careful measurements that transmutation of water to earth was not possible, but that the sediment observed from boiling water came from the container. He burnt phosphorus and sulfur in air, and proved that the products weighed more than the original samples, with the mass gained being lost from the air. Thus, in 1789, he established the Law of
Antoine-Laurent de Lavoisier demonstrated with careful measurements that transmutation of water to earth was not possible, but that the sediment observed from boiling water came from the container. He burnt phosphorus and sulfur in air, and proved that the products weighed more than the original samples, with the mass gained being lost from the air. Thus, in 1789, he established the Law of 
 In 1803, English meteorologist and chemist John Dalton proposed Dalton's law, which describes the relationship between the components in a mixture of gases and the relative pressure each contributes to that of the overall mixture. Discovered in 1801, this concept is also known as Dalton's law of partial pressures.
Dalton also proposed a modern atomic theory in 1803 which stated that all matter was composed of small indivisible particles termed atoms, atoms of a given element possess unique characteristics and weight, and three types of atoms exist: simple (elements), compound (simple molecules), and complex (complex molecules). In 1808, Dalton first published ''New System of Chemical Philosophy'' (1808–1827), in which he outlined the first modern scientific description of the atomic theory. This work identified chemical elements as a specific type of atom, therefore rejecting Isaac Newton, Newton's theory of chemical affinities.
Instead, Dalton inferred proportions of elements in compounds by taking ratios of the weights of reactants, setting the atomic weight of hydrogen to be identically one. Following Jeremias Benjamin Richter (known for introducing the term ''stoichiometry''), he proposed that chemical elements combine in integral ratios. This is known as the law of multiple proportions or Dalton's law, and Dalton included a clear description of the law in his ''New System of Chemical Philosophy''. The law of multiple proportions is one of the basic laws of stoichiometry used to establish the atomic theory. Despite the importance of the work as the first view of atoms as physically real entities and the introduction of a system of chemical symbols, ''New System of Chemical Philosophy'' devoted almost as much space to the caloric theory as to atomism.
French chemist Joseph Proust proposed the law of definite proportions, which states that elements always combine in small, whole number ratios to form compounds, based on several experiments conducted between 1797 and 1804 Along with the law of multiple proportions, the law of definite proportions forms the basis of stoichiometry. The law of definite proportions and constant composition do not prove that atoms exist, but they are difficult to explain without assuming that chemical compounds are formed when atoms combine in constant proportions.
In 1803, English meteorologist and chemist John Dalton proposed Dalton's law, which describes the relationship between the components in a mixture of gases and the relative pressure each contributes to that of the overall mixture. Discovered in 1801, this concept is also known as Dalton's law of partial pressures.
Dalton also proposed a modern atomic theory in 1803 which stated that all matter was composed of small indivisible particles termed atoms, atoms of a given element possess unique characteristics and weight, and three types of atoms exist: simple (elements), compound (simple molecules), and complex (complex molecules). In 1808, Dalton first published ''New System of Chemical Philosophy'' (1808–1827), in which he outlined the first modern scientific description of the atomic theory. This work identified chemical elements as a specific type of atom, therefore rejecting Isaac Newton, Newton's theory of chemical affinities.
Instead, Dalton inferred proportions of elements in compounds by taking ratios of the weights of reactants, setting the atomic weight of hydrogen to be identically one. Following Jeremias Benjamin Richter (known for introducing the term ''stoichiometry''), he proposed that chemical elements combine in integral ratios. This is known as the law of multiple proportions or Dalton's law, and Dalton included a clear description of the law in his ''New System of Chemical Philosophy''. The law of multiple proportions is one of the basic laws of stoichiometry used to establish the atomic theory. Despite the importance of the work as the first view of atoms as physically real entities and the introduction of a system of chemical symbols, ''New System of Chemical Philosophy'' devoted almost as much space to the caloric theory as to atomism.
French chemist Joseph Proust proposed the law of definite proportions, which states that elements always combine in small, whole number ratios to form compounds, based on several experiments conducted between 1797 and 1804 Along with the law of multiple proportions, the law of definite proportions forms the basis of stoichiometry. The law of definite proportions and constant composition do not prove that atoms exist, but they are difficult to explain without assuming that chemical compounds are formed when atoms combine in constant proportions.
 A Swedish chemist and disciple of Dalton, Jöns Jacob Berzelius embarked on a systematic program to try to make accurate and precise quantitative measurements and to ensure the purity of chemicals. Along with Lavoisier, Boyle, and Dalton, Berzelius is known as the father of modern chemistry. In 1828 he compiled a table of relative atomic weights, where
A Swedish chemist and disciple of Dalton, Jöns Jacob Berzelius embarked on a systematic program to try to make accurate and precise quantitative measurements and to ensure the purity of chemicals. Along with Lavoisier, Boyle, and Dalton, Berzelius is known as the father of modern chemistry. In 1828 he compiled a table of relative atomic weights, where  English chemist Humphry Davy was a pioneer in the field of electrolysis, using Alessandro Volta's voltaic pile to split up common compounds and thus isolate a series of new elements. He went on to electrolyse molten salts and discovered several new metals, especially sodium and potassium, highly reactive elements known as the alkali metals. Potassium, the first metal that was isolated by electrolysis, was discovered in 1807 by Davy, who derived it from potassium hydroxide, caustic potash (KOH). Before the 19th century, no distinction was made between potassium and sodium. Sodium was first isolated by Davy in the same year by passing an electric current through molten sodium hydroxide (NaOH). When Davy heard that Berzelius and Pontin prepared calcium amalgam by electrolyzing lime in mercury, he tried it himself. Davy was successful, and discovered calcium in 1808 by electrolyzing a mixture of Lime (material), lime and mercuric oxide. He worked with electrolysis throughout his life and, in 1808, he isolated magnesium, strontium and barium.
Davy also experimented with gases by inhaling them. This experimental procedure nearly proved fatal on several occasions, but led to the discovery of the unusual effects of nitrous oxide, which came to be known as laughing gas. Chlorine was discovered in 1774 by Swedish chemist
English chemist Humphry Davy was a pioneer in the field of electrolysis, using Alessandro Volta's voltaic pile to split up common compounds and thus isolate a series of new elements. He went on to electrolyse molten salts and discovered several new metals, especially sodium and potassium, highly reactive elements known as the alkali metals. Potassium, the first metal that was isolated by electrolysis, was discovered in 1807 by Davy, who derived it from potassium hydroxide, caustic potash (KOH). Before the 19th century, no distinction was made between potassium and sodium. Sodium was first isolated by Davy in the same year by passing an electric current through molten sodium hydroxide (NaOH). When Davy heard that Berzelius and Pontin prepared calcium amalgam by electrolyzing lime in mercury, he tried it himself. Davy was successful, and discovered calcium in 1808 by electrolyzing a mixture of Lime (material), lime and mercuric oxide. He worked with electrolysis throughout his life and, in 1808, he isolated magnesium, strontium and barium.
Davy also experimented with gases by inhaling them. This experimental procedure nearly proved fatal on several occasions, but led to the discovery of the unusual effects of nitrous oxide, which came to be known as laughing gas. Chlorine was discovered in 1774 by Swedish chemist  French chemist Joseph Louis Gay-Lussac shared the interest of Lavoisier and others in the quantitative study of the properties of gases. From his first major program of research in 1801–1802, he concluded that equal volumes of all gases expand equally with the same increase in temperature: this conclusion is usually called "Charles's law", as Gay-Lussac gave credit to Jacques Charles, who had arrived at nearly the same conclusion in the 1780s but had not published it.
French chemist Joseph Louis Gay-Lussac shared the interest of Lavoisier and others in the quantitative study of the properties of gases. From his first major program of research in 1801–1802, he concluded that equal volumes of all gases expand equally with the same increase in temperature: this conclusion is usually called "Charles's law", as Gay-Lussac gave credit to Jacques Charles, who had arrived at nearly the same conclusion in the 1780s but had not published it. After Dalton published his atomic theory in 1808, certain of his central ideas were soon adopted by most chemists. However, uncertainty persisted for half a century about how atomic theory was to be configured and applied to concrete situations; chemists in different countries developed several different incompatible atomistic systems. A paper that suggested a way out of this difficult situation was published as early as 1811 by the Italian physicist Amedeo Avogadro (1776–1856), who hypothesized that equal volumes of gases at the same temperature and
After Dalton published his atomic theory in 1808, certain of his central ideas were soon adopted by most chemists. However, uncertainty persisted for half a century about how atomic theory was to be configured and applied to concrete situations; chemists in different countries developed several different incompatible atomistic systems. A paper that suggested a way out of this difficult situation was published as early as 1811 by the Italian physicist Amedeo Avogadro (1776–1856), who hypothesized that equal volumes of gases at the same temperature and  Avogadro's hypothesis began to gain broad appeal among chemists only after his compatriot and fellow scientist Stanislao Cannizzaro demonstrated its value in 1858, two years after Avogadro's death. Cannizzaro's chemical interests had originally centered on natural products and on reactions of aromatic compounds; in 1853 he discovered that when benzaldehyde is treated with concentrated base, both benzoic acid and benzyl alcohol are produced—a phenomenon known today as the Cannizzaro reaction. In his 1858 pamphlet, Cannizzaro showed that a complete return to the ideas of Avogadro could be used to construct a consistent and robust theoretical structure that fit nearly all of the available empirical evidence. For instance, he pointed to evidence that suggested that not all elementary gases consist of two atoms per molecule—some were monatomic, most were diatomic, and a few were even more complex.
Another point of contention had been the formulas for compounds of the alkali metals (such as sodium) and the alkaline earth metals (such as calcium), which, in view of their striking chemical analogies, most chemists had wanted to assign to the same formula type. Cannizzaro argued that placing these metals in different categories had the beneficial result of eliminating certain anomalies when using their physical properties to deduce atomic weights. Unfortunately, Cannizzaro's pamphlet was published initially only in Italian and had little immediate impact. The real breakthrough came with an Karlsruhe Congress, international chemical congress held in the German town of Karlsruhe in September 1860, at which most of the leading European chemists were present. The Karlsruhe Congress had been arranged by Kekulé, Wurtz, and a few others who shared Cannizzaro's sense of the direction chemistry should go. Speaking in French (as everyone there did), Cannizzaro's eloquence and logic made an indelible impression on the assembled body. Moreover, his friend Angelo Pavesi distributed Cannizzaro's pamphlet to attendees at the end of the meeting; more than one chemist later wrote of the decisive impression the reading of this document provided. For instance, Julius Lothar Meyer, Lothar Meyer later wrote that on reading Cannizzaro's paper, "The scales seemed to fall from my eyes." Cannizzaro thus played a crucial role in winning the battle for reform. The system advocated by him, and soon thereafter adopted by most leading chemists, is substantially identical to what is still used today.
Avogadro's hypothesis began to gain broad appeal among chemists only after his compatriot and fellow scientist Stanislao Cannizzaro demonstrated its value in 1858, two years after Avogadro's death. Cannizzaro's chemical interests had originally centered on natural products and on reactions of aromatic compounds; in 1853 he discovered that when benzaldehyde is treated with concentrated base, both benzoic acid and benzyl alcohol are produced—a phenomenon known today as the Cannizzaro reaction. In his 1858 pamphlet, Cannizzaro showed that a complete return to the ideas of Avogadro could be used to construct a consistent and robust theoretical structure that fit nearly all of the available empirical evidence. For instance, he pointed to evidence that suggested that not all elementary gases consist of two atoms per molecule—some were monatomic, most were diatomic, and a few were even more complex.
Another point of contention had been the formulas for compounds of the alkali metals (such as sodium) and the alkaline earth metals (such as calcium), which, in view of their striking chemical analogies, most chemists had wanted to assign to the same formula type. Cannizzaro argued that placing these metals in different categories had the beneficial result of eliminating certain anomalies when using their physical properties to deduce atomic weights. Unfortunately, Cannizzaro's pamphlet was published initially only in Italian and had little immediate impact. The real breakthrough came with an Karlsruhe Congress, international chemical congress held in the German town of Karlsruhe in September 1860, at which most of the leading European chemists were present. The Karlsruhe Congress had been arranged by Kekulé, Wurtz, and a few others who shared Cannizzaro's sense of the direction chemistry should go. Speaking in French (as everyone there did), Cannizzaro's eloquence and logic made an indelible impression on the assembled body. Moreover, his friend Angelo Pavesi distributed Cannizzaro's pamphlet to attendees at the end of the meeting; more than one chemist later wrote of the decisive impression the reading of this document provided. For instance, Julius Lothar Meyer, Lothar Meyer later wrote that on reading Cannizzaro's paper, "The scales seemed to fall from my eyes." Cannizzaro thus played a crucial role in winning the battle for reform. The system advocated by him, and soon thereafter adopted by most leading chemists, is substantially identical to what is still used today.
 British chemist and physicist William Crookes is noted for his cathode ray studies, fundamental in the development of atomic physics. His researches on electrical discharges through a rarefied gas led him to observe the dark space around the cathode, now called the Crookes dark space. He demonstrated that cathode rays travel in straight lines and produce phosphorescence and heat when they strike certain materials. A pioneer of vacuum tubes, Crookes invented the Crookes tube – an early experimental discharge tube, with partial vacuum with which he studied the behavior of cathode rays. With the introduction of Spectroscopy, spectrum analysis by Robert Bunsen and Gustav Kirchhoff (1859–1860), Crookes applied the new technique to the study of selenium compounds. Bunsen and Kirchhoff had previously used spectroscopy as a means of chemical analysis to discover caesium and rubidium. In 1861, Crookes used this process to discover thallium in some seleniferous deposits. He continued work on that new element, isolated it, studied its properties, and in 1873 determined its atomic weight. During his studies of thallium, Crookes discovered the principle of the Crookes radiometer, a device that converts light radiation into rotary motion. The principle of this radiometer has found numerous applications in the development of sensitive measuring instruments.
In 1862, Alexander Parkes exhibited Parkesine, one of the earliest synthetic polymers, at the International Exhibition in London. This discovery formed the foundation of the modern plastics industry. In 1864, Cato Maximilian Guldberg and Peter Waage, building on Claude Louis Berthollet's ideas, proposed the law of mass action. In 1865, Johann Josef Loschmidt determined the number of molecules in a mole (unit), mole, later named Avogadro constant, Avogadro's number.
In 1865, August Kekulé, based partially on the work of Loschmidt and others, established the structure of benzene as a six carbon ring with alternating single and double bonds. Kekulé's novel proposal for benzene's cyclic structure was much contested but was never replaced by a superior theory. This theory provided the scientific basis for the dramatic expansion of the German chemical industry in the last third of the 19th century. Kekulé is also famous for having clarified the nature of aromatic compounds, which are compounds based on the benzene molecule. In 1865, Adolf von Baeyer began work on indigo dye, a milestone in modern industrial organic chemistry which revolutionized the dye industry.
Swedish chemist and inventor Alfred Nobel found that when nitroglycerin was incorporated in an absorbent inert substance like ''kieselguhr'' (diatomaceous earth) it became safer and more convenient to handle, and this mixture he patented in 1867 as dynamite. Nobel later on combined nitroglycerin with various nitrocellulose compounds, similar to collodion, but settled on a more efficient recipe combining another nitrate explosive, and obtained a transparent, jelly-like substance, which was a more powerful explosive than dynamite. Gelignite, or blasting gelatin, as it was named, was patented in 1876; and was followed by a host of similar combinations, modified by the addition of potassium nitrate and various other substances.
British chemist and physicist William Crookes is noted for his cathode ray studies, fundamental in the development of atomic physics. His researches on electrical discharges through a rarefied gas led him to observe the dark space around the cathode, now called the Crookes dark space. He demonstrated that cathode rays travel in straight lines and produce phosphorescence and heat when they strike certain materials. A pioneer of vacuum tubes, Crookes invented the Crookes tube – an early experimental discharge tube, with partial vacuum with which he studied the behavior of cathode rays. With the introduction of Spectroscopy, spectrum analysis by Robert Bunsen and Gustav Kirchhoff (1859–1860), Crookes applied the new technique to the study of selenium compounds. Bunsen and Kirchhoff had previously used spectroscopy as a means of chemical analysis to discover caesium and rubidium. In 1861, Crookes used this process to discover thallium in some seleniferous deposits. He continued work on that new element, isolated it, studied its properties, and in 1873 determined its atomic weight. During his studies of thallium, Crookes discovered the principle of the Crookes radiometer, a device that converts light radiation into rotary motion. The principle of this radiometer has found numerous applications in the development of sensitive measuring instruments.
In 1862, Alexander Parkes exhibited Parkesine, one of the earliest synthetic polymers, at the International Exhibition in London. This discovery formed the foundation of the modern plastics industry. In 1864, Cato Maximilian Guldberg and Peter Waage, building on Claude Louis Berthollet's ideas, proposed the law of mass action. In 1865, Johann Josef Loschmidt determined the number of molecules in a mole (unit), mole, later named Avogadro constant, Avogadro's number.
In 1865, August Kekulé, based partially on the work of Loschmidt and others, established the structure of benzene as a six carbon ring with alternating single and double bonds. Kekulé's novel proposal for benzene's cyclic structure was much contested but was never replaced by a superior theory. This theory provided the scientific basis for the dramatic expansion of the German chemical industry in the last third of the 19th century. Kekulé is also famous for having clarified the nature of aromatic compounds, which are compounds based on the benzene molecule. In 1865, Adolf von Baeyer began work on indigo dye, a milestone in modern industrial organic chemistry which revolutionized the dye industry.
Swedish chemist and inventor Alfred Nobel found that when nitroglycerin was incorporated in an absorbent inert substance like ''kieselguhr'' (diatomaceous earth) it became safer and more convenient to handle, and this mixture he patented in 1867 as dynamite. Nobel later on combined nitroglycerin with various nitrocellulose compounds, similar to collodion, but settled on a more efficient recipe combining another nitrate explosive, and obtained a transparent, jelly-like substance, which was a more powerful explosive than dynamite. Gelignite, or blasting gelatin, as it was named, was patented in 1876; and was followed by a host of similar combinations, modified by the addition of potassium nitrate and various other substances.
 An important breakthrough in making sense of the list of known chemical elements (as well as in understanding the internal structure of atoms) was Dmitri Mendeleev's development of the first modern periodic table, or the periodic classification of the elements. Mendeleev, a Russian chemist, felt that there was some type of order to the elements and he spent more than thirteen years of his life collecting data and assembling the concept, initially with the idea of resolving some of the disorder in the field for his students. Mendeleev found that, when all the known chemical elements were arranged in order of increasing atomic weight, the resulting table displayed a recurring pattern, or periodicity, of properties within groups of elements. Mendeleev's law allowed him to build up a systematic periodic table of all the 66 elements then known based on atomic mass, which he published in ''Principles of Chemistry'' in 1869. His first Periodic Table was compiled on the basis of arranging the elements in ascending order of atomic weight and grouping them by similarity of properties.
Mendeleev had such faith in the validity of the periodic law that he proposed changes to the generally accepted values for the atomic weight of a few elements and, in his version of the periodic table of 1871, predicted the locations within the table of unknown elements together with their properties. He even predicted the likely properties of three yet-to-be-discovered elements, which he called Mendeleev's predicted elements, ekaboron (Eb), ekaaluminium (Ea), and ekasilicon (Es), which proved to be good predictors of the properties of scandium, gallium, and germanium, respectively, which each fill the spot in the periodic table assigned by Mendeleev.
At first the periodic system did not raise interest among chemists. However, with the discovery of the predicted elements, notably gallium in 1875, scandium in 1879, and germanium in 1886, it began to win wide acceptance. The subsequent proof of many of his predictions within his lifetime brought fame to Mendeleev as the founder of the periodic law. This organization surpassed earlier attempts at classification by Alexandre-Émile Béguyer de Chancourtois, who published the telluric helix, an early, three-dimensional version of the periodic table of the elements in 1862, John Alexander Reina Newlands, John Newlands, who proposed the law of octaves (a precursor to the periodic law) in 1864, and Julius Lothar Meyer, Lothar Meyer, who developed an early version of the periodic table with 28 elements organized by valence (chemistry), valence in 1864. Mendeleev's table did not include any of the noble gases, however, which had not yet been discovered. Gradually the periodic law and table became the framework for a great part of chemical theory. By the time Mendeleev died in 1907, he enjoyed international recognition and had received distinctions and awards from many countries.
In 1873, Jacobus Henricus van 't Hoff and Joseph Achille Le Bel, working independently, developed a model of chemical bonds, chemical bonding that explained the chirality experiments of Pasteur and provided a physical cause for optical activity in chiral compounds. van 't Hoff's publication, called V''oorstel tot Uitbreiding der Tegenwoordige in de Scheikunde gebruikte Structuurformules in de Ruimte'', etc. (Proposal for the development of 3-dimensional chemical structural formulae) and consisting of twelve pages of text and one page of diagrams, gave the impetus to the development of stereochemistry. The concept of the "asymmetrical carbon atom", dealt with in this publication, supplied an explanation of the occurrence of numerous isomers, inexplicable by means of the then current structural formulae. At the same time he pointed out the existence of relationship between optical activity and the presence of an asymmetrical carbon atom.
An important breakthrough in making sense of the list of known chemical elements (as well as in understanding the internal structure of atoms) was Dmitri Mendeleev's development of the first modern periodic table, or the periodic classification of the elements. Mendeleev, a Russian chemist, felt that there was some type of order to the elements and he spent more than thirteen years of his life collecting data and assembling the concept, initially with the idea of resolving some of the disorder in the field for his students. Mendeleev found that, when all the known chemical elements were arranged in order of increasing atomic weight, the resulting table displayed a recurring pattern, or periodicity, of properties within groups of elements. Mendeleev's law allowed him to build up a systematic periodic table of all the 66 elements then known based on atomic mass, which he published in ''Principles of Chemistry'' in 1869. His first Periodic Table was compiled on the basis of arranging the elements in ascending order of atomic weight and grouping them by similarity of properties.
Mendeleev had such faith in the validity of the periodic law that he proposed changes to the generally accepted values for the atomic weight of a few elements and, in his version of the periodic table of 1871, predicted the locations within the table of unknown elements together with their properties. He even predicted the likely properties of three yet-to-be-discovered elements, which he called Mendeleev's predicted elements, ekaboron (Eb), ekaaluminium (Ea), and ekasilicon (Es), which proved to be good predictors of the properties of scandium, gallium, and germanium, respectively, which each fill the spot in the periodic table assigned by Mendeleev.
At first the periodic system did not raise interest among chemists. However, with the discovery of the predicted elements, notably gallium in 1875, scandium in 1879, and germanium in 1886, it began to win wide acceptance. The subsequent proof of many of his predictions within his lifetime brought fame to Mendeleev as the founder of the periodic law. This organization surpassed earlier attempts at classification by Alexandre-Émile Béguyer de Chancourtois, who published the telluric helix, an early, three-dimensional version of the periodic table of the elements in 1862, John Alexander Reina Newlands, John Newlands, who proposed the law of octaves (a precursor to the periodic law) in 1864, and Julius Lothar Meyer, Lothar Meyer, who developed an early version of the periodic table with 28 elements organized by valence (chemistry), valence in 1864. Mendeleev's table did not include any of the noble gases, however, which had not yet been discovered. Gradually the periodic law and table became the framework for a great part of chemical theory. By the time Mendeleev died in 1907, he enjoyed international recognition and had received distinctions and awards from many countries.
In 1873, Jacobus Henricus van 't Hoff and Joseph Achille Le Bel, working independently, developed a model of chemical bonds, chemical bonding that explained the chirality experiments of Pasteur and provided a physical cause for optical activity in chiral compounds. van 't Hoff's publication, called V''oorstel tot Uitbreiding der Tegenwoordige in de Scheikunde gebruikte Structuurformules in de Ruimte'', etc. (Proposal for the development of 3-dimensional chemical structural formulae) and consisting of twelve pages of text and one page of diagrams, gave the impetus to the development of stereochemistry. The concept of the "asymmetrical carbon atom", dealt with in this publication, supplied an explanation of the occurrence of numerous isomers, inexplicable by means of the then current structural formulae. At the same time he pointed out the existence of relationship between optical activity and the presence of an asymmetrical carbon atom.
 American mathematical physicist Josiah Willard Gibbs, J. Willard Gibbs's work on the applications of thermodynamics was instrumental in transforming physical chemistry into a rigorous deductive science. During the years from 1876 to 1878, Gibbs worked on the principles of thermodynamics, applying them to the complex processes involved in chemical reactions. He discovered the concept of chemical potential, or the "fuel" that makes chemical reactions work. In 1876 he published his most famous contribution, "On the Equilibrium of Heterogeneous Substances", a compilation of his work on thermodynamics and physical chemistry which laid out the concept of Thermodynamic free energy, free energy to explain the physical basis of chemical equilibria. In these essays were the beginnings of Gibbs’ theories of phases of matter: he considered each state of matter a phase, and each substance a component. Gibbs took all of the variables involved in a chemical reaction – temperature, pressure, energy, volume, and entropy – and included them in one simple equation known as Gibbs' phase rule.
Within this paper was perhaps his most outstanding contribution, the introduction of the concept of free energy, now universally called Gibbs free energy in his honor. The Gibbs free energy relates the tendency of a physical or chemical system to simultaneously lower its energy and increase its disorder, or entropy, in a spontaneous natural process. Gibbs's approach allows a researcher to calculate the change in free energy in the process, such as in a chemical reaction, and how fast it will happen. Since virtually all chemical processes and many physical ones involve such changes, his work has significantly impacted both the theoretical and experiential aspects of these sciences. In 1877, Ludwig Boltzmann established statistical derivations of many important physical and chemical concepts, including entropy, and distributions of molecular velocities in the gas phase. Together with Boltzmann and James Clerk Maxwell, Gibbs created a new branch of theoretical physics called statistical mechanics (a term that he coined), explaining the laws of thermodynamics as consequences of the statistical properties of large ensembles of particles. Gibbs also worked on the application of Maxwell's equations to problems in physical optics. Gibbs's derivation of the phenomenological laws of thermodynamics from the statistical properties of systems with many particles was presented in his highly influential textbook ''Elementary Principles in Statistical Mechanics'', published in 1902, a year before his death. In that work, Gibbs reviewed the relationship between the laws of thermodynamics and the statistical theory of molecular motions. The overshooting of the original function by partial sums of Fourier series at points of discontinuity is known as the Gibbs phenomenon.
American mathematical physicist Josiah Willard Gibbs, J. Willard Gibbs's work on the applications of thermodynamics was instrumental in transforming physical chemistry into a rigorous deductive science. During the years from 1876 to 1878, Gibbs worked on the principles of thermodynamics, applying them to the complex processes involved in chemical reactions. He discovered the concept of chemical potential, or the "fuel" that makes chemical reactions work. In 1876 he published his most famous contribution, "On the Equilibrium of Heterogeneous Substances", a compilation of his work on thermodynamics and physical chemistry which laid out the concept of Thermodynamic free energy, free energy to explain the physical basis of chemical equilibria. In these essays were the beginnings of Gibbs’ theories of phases of matter: he considered each state of matter a phase, and each substance a component. Gibbs took all of the variables involved in a chemical reaction – temperature, pressure, energy, volume, and entropy – and included them in one simple equation known as Gibbs' phase rule.
Within this paper was perhaps his most outstanding contribution, the introduction of the concept of free energy, now universally called Gibbs free energy in his honor. The Gibbs free energy relates the tendency of a physical or chemical system to simultaneously lower its energy and increase its disorder, or entropy, in a spontaneous natural process. Gibbs's approach allows a researcher to calculate the change in free energy in the process, such as in a chemical reaction, and how fast it will happen. Since virtually all chemical processes and many physical ones involve such changes, his work has significantly impacted both the theoretical and experiential aspects of these sciences. In 1877, Ludwig Boltzmann established statistical derivations of many important physical and chemical concepts, including entropy, and distributions of molecular velocities in the gas phase. Together with Boltzmann and James Clerk Maxwell, Gibbs created a new branch of theoretical physics called statistical mechanics (a term that he coined), explaining the laws of thermodynamics as consequences of the statistical properties of large ensembles of particles. Gibbs also worked on the application of Maxwell's equations to problems in physical optics. Gibbs's derivation of the phenomenological laws of thermodynamics from the statistical properties of systems with many particles was presented in his highly influential textbook ''Elementary Principles in Statistical Mechanics'', published in 1902, a year before his death. In that work, Gibbs reviewed the relationship between the laws of thermodynamics and the statistical theory of molecular motions. The overshooting of the original function by partial sums of Fourier series at points of discontinuity is known as the Gibbs phenomenon.
 The following year, Ramsay liberated another inert gas from a mineral called cleveite; this proved to be helium, previously known only in the solar spectrum. In his book ''The Gases of the Atmosphere'' (1896), Ramsay showed that the positions of helium and argon in the periodic table of elements indicated that at least three more noble gases might exist. In 1898 Ramsay and the British chemist Morris Travers, Morris W. Travers isolated these elements—called neon, krypton, and xenon—from air and brought them to a liquid state at low temperature and high pressure. Sir William Ramsay worked with Frederick Soddy to demonstrate, in 1903, that alpha particles (helium nuclei) were continually produced during the radioactive decay of a sample of radium. Ramsay was awarded the 1904 Nobel Prize for Chemistry in recognition of "services in the discovery of the inert gaseous elements in the air, and his determination of their place in the periodic system."
In 1897, J. J. Thomson discovered the electron using the cathode ray tube. In 1898, Wilhelm Wien demonstrated that canal rays (streams of positive ions) can be deflected by magnetic fields and that the amount of deflection is proportional to the mass-to-charge ratio. This discovery would lead to the analytical chemistry, analytical technique known as mass spectrometry in 1912.
The following year, Ramsay liberated another inert gas from a mineral called cleveite; this proved to be helium, previously known only in the solar spectrum. In his book ''The Gases of the Atmosphere'' (1896), Ramsay showed that the positions of helium and argon in the periodic table of elements indicated that at least three more noble gases might exist. In 1898 Ramsay and the British chemist Morris Travers, Morris W. Travers isolated these elements—called neon, krypton, and xenon—from air and brought them to a liquid state at low temperature and high pressure. Sir William Ramsay worked with Frederick Soddy to demonstrate, in 1903, that alpha particles (helium nuclei) were continually produced during the radioactive decay of a sample of radium. Ramsay was awarded the 1904 Nobel Prize for Chemistry in recognition of "services in the discovery of the inert gaseous elements in the air, and his determination of their place in the periodic system."
In 1897, J. J. Thomson discovered the electron using the cathode ray tube. In 1898, Wilhelm Wien demonstrated that canal rays (streams of positive ions) can be deflected by magnetic fields and that the amount of deflection is proportional to the mass-to-charge ratio. This discovery would lead to the analytical chemistry, analytical technique known as mass spectrometry in 1912.
 Marie Curie, Marie Skłodowska-Curie was a Polish-born French physicist and chemist who is famous for her pioneering research on radioactive decay, radioactivity. She and her husband are considered to have laid the cornerstone of the nuclear age with their research on radioactivity. Marie was fascinated with the work of Henri Becquerel, a French physicist who discovered in 1896 that uranium casts off rays similar to the X-rays discovered by Wilhelm Röntgen. Marie Curie began studying uranium in late 1897 and theorized, according to a 1904 article she wrote for Century magazine, "that the emission of rays by the compounds of uranium is a property of the metal itself—that it is an atomic property of the element uranium independent of its chemical or physical state." Curie took Becquerel's work a few steps further, conducting her own experiments on uranium rays. She discovered that the rays remained constant, no matter the condition or form of the uranium. The rays, she theorized, came from the element's atomic structure. This revolutionary idea created the field of atomic physics and the Curies coined the word ''radioactivity'' to describe the phenomena.
Marie Curie, Marie Skłodowska-Curie was a Polish-born French physicist and chemist who is famous for her pioneering research on radioactive decay, radioactivity. She and her husband are considered to have laid the cornerstone of the nuclear age with their research on radioactivity. Marie was fascinated with the work of Henri Becquerel, a French physicist who discovered in 1896 that uranium casts off rays similar to the X-rays discovered by Wilhelm Röntgen. Marie Curie began studying uranium in late 1897 and theorized, according to a 1904 article she wrote for Century magazine, "that the emission of rays by the compounds of uranium is a property of the metal itself—that it is an atomic property of the element uranium independent of its chemical or physical state." Curie took Becquerel's work a few steps further, conducting her own experiments on uranium rays. She discovered that the rays remained constant, no matter the condition or form of the uranium. The rays, she theorized, came from the element's atomic structure. This revolutionary idea created the field of atomic physics and the Curies coined the word ''radioactivity'' to describe the phenomena.
 Pierre and Marie further explored radioactivity by working to separate the substances in uranium ores and then using the electrometer to make radiation measurements to ‘trace’ the minute amount of unknown radioactive element among the fractions that resulted. Working with the mineral Uraninite, pitchblende, the pair discovered a new radioactive element in 1898. They named the element polonium, after Marie's native country of Poland. On December 21, 1898, the Curies detected the presence of another radioactive material in the pitchblende. They presented this finding to the French Academy of Sciences on December 26, proposing that the new element be called radium. The Curies then went to work isolating polonium and radium from naturally occurring compounds to prove that they were new elements. In 1902, the Curies announced that they had produced a decigram of pure radium, demonstrating its existence as a unique chemical element. While it took three years for them to isolate radium, they were never able to isolate polonium. Along with the discovery of two new elements and finding techniques for isolating radioactive isotopes, Curie oversaw the world's first studies into the treatment of neoplasms, using radioactive isotopes. With Henri Becquerel and her husband, Pierre Curie, she was awarded the 1903 Nobel Prize for Physics. She was the sole winner of the 1911 Nobel Prize for Chemistry. She was the first woman to win a Nobel Prize, and she is the only woman to win the award in two different fields.
While working with Marie to extract pure substances from ores, an undertaking that really required industrial resources but that they achieved in relatively primitive conditions, Pierre himself concentrated on the physical study (including luminous and chemical effects) of the new radiations. Through the action of magnetic fields on the rays given out by the radium, he proved the existence of particles that were electrically positive, negative, and neutral; these Ernest Rutherford was afterward to call alpha, beta, and gamma rays. Pierre then studied these radiations by calorimetry and also observed the physiological effects of radium, thus opening the way to radium therapy. Among Pierre Curie's discoveries were that ferromagnetic substances exhibited a critical temperature transition, above which the substances lost their ferromagnetic behavior – this is known as the "Curie temperature, Curie point." He was elected to the Academy of Sciences (1905), having in 1903 jointly with Marie received the Royal Society's prestigious Davy Medal and jointly with her and Becquerel the Nobel Prize for Physics. He was run over by a carriage in the rue Dauphine in Paris in 1906 and died instantly. His complete works were published in 1908.
Pierre and Marie further explored radioactivity by working to separate the substances in uranium ores and then using the electrometer to make radiation measurements to ‘trace’ the minute amount of unknown radioactive element among the fractions that resulted. Working with the mineral Uraninite, pitchblende, the pair discovered a new radioactive element in 1898. They named the element polonium, after Marie's native country of Poland. On December 21, 1898, the Curies detected the presence of another radioactive material in the pitchblende. They presented this finding to the French Academy of Sciences on December 26, proposing that the new element be called radium. The Curies then went to work isolating polonium and radium from naturally occurring compounds to prove that they were new elements. In 1902, the Curies announced that they had produced a decigram of pure radium, demonstrating its existence as a unique chemical element. While it took three years for them to isolate radium, they were never able to isolate polonium. Along with the discovery of two new elements and finding techniques for isolating radioactive isotopes, Curie oversaw the world's first studies into the treatment of neoplasms, using radioactive isotopes. With Henri Becquerel and her husband, Pierre Curie, she was awarded the 1903 Nobel Prize for Physics. She was the sole winner of the 1911 Nobel Prize for Chemistry. She was the first woman to win a Nobel Prize, and she is the only woman to win the award in two different fields.
While working with Marie to extract pure substances from ores, an undertaking that really required industrial resources but that they achieved in relatively primitive conditions, Pierre himself concentrated on the physical study (including luminous and chemical effects) of the new radiations. Through the action of magnetic fields on the rays given out by the radium, he proved the existence of particles that were electrically positive, negative, and neutral; these Ernest Rutherford was afterward to call alpha, beta, and gamma rays. Pierre then studied these radiations by calorimetry and also observed the physiological effects of radium, thus opening the way to radium therapy. Among Pierre Curie's discoveries were that ferromagnetic substances exhibited a critical temperature transition, above which the substances lost their ferromagnetic behavior – this is known as the "Curie temperature, Curie point." He was elected to the Academy of Sciences (1905), having in 1903 jointly with Marie received the Royal Society's prestigious Davy Medal and jointly with her and Becquerel the Nobel Prize for Physics. He was run over by a carriage in the rue Dauphine in Paris in 1906 and died instantly. His complete works were published in 1908.
 New Zealand-born chemist and physicist Ernest Rutherford is considered to be "the father of nuclear physics." Rutherford is best known for devising the names alpha particle, alpha, beta particle, beta, and gamma ray, gamma to classify various forms of radioactive "rays" which were poorly understood at his time (alpha and beta rays are particle beams, while gamma rays are a form of high-energy electromagnetic radiation). Rutherford deflected alpha rays with both electric and magnetic fields in 1903. Working with Frederick Soddy, Rutherford explained that radioactivity is due to the nuclear transmutation, transmutation of elements, now known to involve nuclear reactions.
New Zealand-born chemist and physicist Ernest Rutherford is considered to be "the father of nuclear physics." Rutherford is best known for devising the names alpha particle, alpha, beta particle, beta, and gamma ray, gamma to classify various forms of radioactive "rays" which were poorly understood at his time (alpha and beta rays are particle beams, while gamma rays are a form of high-energy electromagnetic radiation). Rutherford deflected alpha rays with both electric and magnetic fields in 1903. Working with Frederick Soddy, Rutherford explained that radioactivity is due to the nuclear transmutation, transmutation of elements, now known to involve nuclear reactions.
 In 1903, Mikhail Tsvet invented chromatography, an important analytic technique. In 1904, Hantaro Nagaoka proposed an early nuclear model of the atom, where electrons orbit a dense massive nucleus. In 1905, Fritz Haber and Carl Bosch developed the Haber process for making
In 1903, Mikhail Tsvet invented chromatography, an important analytic technique. In 1904, Hantaro Nagaoka proposed an early nuclear model of the atom, where electrons orbit a dense massive nucleus. In 1905, Fritz Haber and Carl Bosch developed the Haber process for making  In 1905, Albert Einstein explained Brownian motion in a way that definitively proved atomic theory. Leo Baekeland invented bakelite, one of the first commercially successful plastics. In 1909, American physicist Robert Andrews Millikan – who had studied in Europe under Walther Nernst and Max Planck – measured the charge of individual electrons with unprecedented accuracy through the oil drop experiment, in which he measured the electric charges on tiny falling water (and later oil) droplets. His study established that any particular droplet's electrical charge is a multiple of a definite, fundamental value — the electron's charge — and thus a confirmation that all electrons have the same charge and mass. Beginning in 1912, he spent several years investigating and finally proving Albert Einstein's proposed linear relationship between energy and frequency, and providing the first direct Photoelectric effect, photoelectric support for Planck's constant. In 1923 Millikan was awarded the Nobel Prize for Physics.
In 1909, S. P. L. Sørensen invented the pH concept and developed methods for measuring acidity. In 1911, Antonius Van den Broek proposed the idea that the elements on the periodic table are more properly organized by positive nuclear charge rather than atomic weight. In 1911, the first Solvay Conference was held in Brussels, bringing together most of the most prominent scientists of the day. In 1912, William Henry Bragg and William Lawrence Bragg proposed Bragg's law and established the field of X-ray crystallography, an important tool for elucidating the crystal structure of substances. In 1912, Peter Debye used the concept of a molecular dipole to describe asymmetric charge distribution in some molecules.
In 1905, Albert Einstein explained Brownian motion in a way that definitively proved atomic theory. Leo Baekeland invented bakelite, one of the first commercially successful plastics. In 1909, American physicist Robert Andrews Millikan – who had studied in Europe under Walther Nernst and Max Planck – measured the charge of individual electrons with unprecedented accuracy through the oil drop experiment, in which he measured the electric charges on tiny falling water (and later oil) droplets. His study established that any particular droplet's electrical charge is a multiple of a definite, fundamental value — the electron's charge — and thus a confirmation that all electrons have the same charge and mass. Beginning in 1912, he spent several years investigating and finally proving Albert Einstein's proposed linear relationship between energy and frequency, and providing the first direct Photoelectric effect, photoelectric support for Planck's constant. In 1923 Millikan was awarded the Nobel Prize for Physics.
In 1909, S. P. L. Sørensen invented the pH concept and developed methods for measuring acidity. In 1911, Antonius Van den Broek proposed the idea that the elements on the periodic table are more properly organized by positive nuclear charge rather than atomic weight. In 1911, the first Solvay Conference was held in Brussels, bringing together most of the most prominent scientists of the day. In 1912, William Henry Bragg and William Lawrence Bragg proposed Bragg's law and established the field of X-ray crystallography, an important tool for elucidating the crystal structure of substances. In 1912, Peter Debye used the concept of a molecular dipole to describe asymmetric charge distribution in some molecules.
 In 1913, Niels Bohr, a Danish physicist, introduced the concepts of quantum mechanics to atomic structure by proposing what is now known as the Bohr model of the atom, where electrons exist only in strictly defined circular orbits around the nucleus similar to rungs on a ladder. The Bohr Model is a planetary model in which the negatively charged electrons orbit a small, positively charged nucleus similar to the planets orbiting the Sun (except that the orbits are not planar) – the gravitational force of the solar system is mathematically akin to the attractive Coulomb (electrical) force between the positively charged nucleus and the negatively charged electrons.
In the Bohr model, however, electrons orbit the nucleus in orbits that have a set size and energy – the energy levels are said to be ''quantized'', which means that only certain orbits with certain radii are allowed; orbits in between simply don't exist. The energy of the orbit is related to its size – that is, the lowest energy is found in the smallest orbit. Bohr also postulated that electromagnetic radiation is absorbed or emitted when an electron moves from one orbit to another. Because only certain electron orbits are permitted, the emission of light accompanying a jump of an electron from an excited energy state to ground state produces a unique emission spectrum for each element. Bohr later received the Nobel Prize in physics for this work.
Niels Bohr also worked on the principle of Complementarity (physics), complementarity, which states that an electron can be interpreted in two mutually exclusive and valid ways. Electrons can be interpreted as wave or particle models. His hypothesis was that an incoming particle would strike the nucleus and create an excited compound nucleus. This formed the basis of his Semi-empirical mass formula, liquid drop model and later provided a theory base for nuclear fission after its discovery of nuclear fission, discovery by chemists Otto Hahn and Fritz Strassman, and explanation and naming by physicists Lise Meitner and Otto Frisch.
In 1913, Niels Bohr, a Danish physicist, introduced the concepts of quantum mechanics to atomic structure by proposing what is now known as the Bohr model of the atom, where electrons exist only in strictly defined circular orbits around the nucleus similar to rungs on a ladder. The Bohr Model is a planetary model in which the negatively charged electrons orbit a small, positively charged nucleus similar to the planets orbiting the Sun (except that the orbits are not planar) – the gravitational force of the solar system is mathematically akin to the attractive Coulomb (electrical) force between the positively charged nucleus and the negatively charged electrons.
In the Bohr model, however, electrons orbit the nucleus in orbits that have a set size and energy – the energy levels are said to be ''quantized'', which means that only certain orbits with certain radii are allowed; orbits in between simply don't exist. The energy of the orbit is related to its size – that is, the lowest energy is found in the smallest orbit. Bohr also postulated that electromagnetic radiation is absorbed or emitted when an electron moves from one orbit to another. Because only certain electron orbits are permitted, the emission of light accompanying a jump of an electron from an excited energy state to ground state produces a unique emission spectrum for each element. Bohr later received the Nobel Prize in physics for this work.
Niels Bohr also worked on the principle of Complementarity (physics), complementarity, which states that an electron can be interpreted in two mutually exclusive and valid ways. Electrons can be interpreted as wave or particle models. His hypothesis was that an incoming particle would strike the nucleus and create an excited compound nucleus. This formed the basis of his Semi-empirical mass formula, liquid drop model and later provided a theory base for nuclear fission after its discovery of nuclear fission, discovery by chemists Otto Hahn and Fritz Strassman, and explanation and naming by physicists Lise Meitner and Otto Frisch.
 In 1913, Henry Moseley, working from Van den Broek's earlier idea, introduced the concept of atomic number to fix some inadequacies of Mendeleev's periodic table, which had been based on atomic weight. The peak of Frederick Soddy's career in radiochemistry was in 1913 with his formulation of the concept of isotopes, which stated that certain elements exist in two or more forms which have different atomic weights but which are indistinguishable chemically. He is remembered for proving the existence of isotopes of certain radioactive elements, and is also credited, along with others, with the discovery of the element protactinium in 1917. In 1913, J. J. Thomson expanded on the work of Wien by showing that charged subatomic particles can be separated by their mass-to-charge ratio, a technique known as mass spectrometry.
In 1913, Henry Moseley, working from Van den Broek's earlier idea, introduced the concept of atomic number to fix some inadequacies of Mendeleev's periodic table, which had been based on atomic weight. The peak of Frederick Soddy's career in radiochemistry was in 1913 with his formulation of the concept of isotopes, which stated that certain elements exist in two or more forms which have different atomic weights but which are indistinguishable chemically. He is remembered for proving the existence of isotopes of certain radioactive elements, and is also credited, along with others, with the discovery of the element protactinium in 1917. In 1913, J. J. Thomson expanded on the work of Wien by showing that charged subatomic particles can be separated by their mass-to-charge ratio, a technique known as mass spectrometry.